Mycoplasma Testing Market Overview and Key Insights:
The global mycoplasma testing market size reached USD 866.6 million in 2024 and is expected to register a revenue CAGR of 9.3% during the forecast period. Growth in monoclonal antibody and vaccine production is a major factor driving market revenue growth. Mycoplasma testing is increasing in monoclonal antibody and vaccine production for ensuring cell culture integrity, regulatory compliance, and product safety throughout manufacturing processes.

According to the World Health Organization (WHO), as of mid-February 2025, data from global sentinel surveillance sites indicate a rising trend in SARS-CoV-2 activity. The test positivity rate has climbed to 11%, marking the highest level recorded since July 2024. Also, as of December 23rd, 2023, only 32% of the population has received at least one COVID-19 vaccine booster dose.
Mycoplasma contamination poses a significant threat to cell culture integrity and can compromise the quality and efficacy of final therapeutic products. Regulatory authorities mandate rigorous mycoplasma testing across all stages of biologics development and manufacturing.
Market Drivers:
Mycoplasma contamination is a prevalent issue in cell culture systems in life science research and the pharmaceutical industry, compromising data validity and results. Maintaining contamination-free cell cultures is crucial for cell-based research and consumer health-conscious manufacturers, requiring mycoplasma testing. Increasing concerns about contamination in cell cultures, the growth of the biotechnology and pharmaceutical industries, and rising pharmaceutical and Research and Development (R&D) investments are contributing significantly to market growth.
According to the National Institute of Health (NIH), Chronic obstructive pulmonary disease (COPD) affects 1 in 10 of the adult global population and is one of the three commonest causes of death worldwide. It is also a major cause of global inequalities in health and is more prevalent where such inequalities are more extreme. Over 10% of cell cultures are contaminated with Mycoplasma species, emphasizing the need for routine testing drives the market expansion.
Market Opportunity:
Technological advancement in mycoplasma testing and detection kits is expected to accelerate market revenue growth over the forecast period. These kits are essential for laboratories to detect and prevent contamination of cell cultures. Common methods include PCR, qPCR, ELISA, and metabolic enzyme activity assays. Routine testing is crucial, considering equipment, regulatory requirements, and desired throughput.
Research and Development Systems has developed the MycoProbe Mycoplasma Detection Assay, a highly sensitive tool for routine screening of mycoplasma contamination in cultured cells. This assay detects Mycoplasma 16S ribosomal RNA (rRNA) with sensitivity comparable to PCR, is compatible with high-throughput screening, and generates results in 4.5 hours without requiring antibiotic-free culture.
Market Trends:
Increasing adoption of mycoplasma testing to detect eight mycoplasma species is emerging as a major trend in the mycoplasma testing market. The MycoSEQ Mycoplasma Detection System is an integrated real-time PCR solution for rapid mycoplasma testing. It requires no live mycoplasma for testing or process validation, offers a flexible approach, and detects all recommended species in European, US, and Japanese Pharmacopeias. Single mycoplasma contamination rates range from 15-35%, while multiple infections can be 7-60%. Advancements in instruments will drive market revenue growth during the forecast period.
Restraints & Challenges:
Complex regulatory landscape, such as Food and Drug Administration (FDA) approval, for example, the U.S. FDA had removed a regulation requiring a specific test for Mycoplasma in live virus vaccines from in vitro living cell cultures. This move is seen as overly restrictive, as other methods are currently used. The removal allows for flexibility in accommodating new technology without compromising public health protections. Gold standard testing requires a 28-day agar and broth cultivation method, which is impractical for short shelf-life biologics.
A study evaluated five commercial molecular assays, including MycoSEQ, MycoTOOL, VenorGEM, ATCC, and BioFire, using 10 cultured Mollicutes spp. The Biofire assay was most sensitive, followed by MycoSEQ and MycoTOOL. Not all assays met the ≤10 CFU/mL LOD requirement, and no interference was observed in the presence of TILs. The scarcity of skilled lab technicians and the adoption of mycoplasma testing kits hinders market growth.
Component Segment Insights and Analysis:
Based on the Component, the Mycoplasma Testing Market is segmented into Kits & Reagents, Instruments, and Services.
Kits & Reagents contributed the largest revenue share in 2024. The Kits & Reagents segment will be sub-segmented into the key areas of Polymerase chain reaction (PCR) Assays, Nucleic Acid Detection Kits, ELISA (Enzyme-Linked Immunosorbent Assay) Kits, Staining, and Others. PCR detects an array of Mycoplasma species with high specificity and efficiency. The key benefits of such a technique are its immediate detection capabilities and simple use.
The LookOut Mycoplasma PCR Detection Kit uses PCR for high sensitivity in detecting Mycoplasma and Acholeplasma contamination in cell cultures and biologicals. It is suitable for UNG pretreatment and direct testing of cell cultures and biologicals. A new PCR assay has been proposed for Mycoplasma detection, demonstrating 100% sensitivity and detecting a minimum of 10 CFU/mL for nine species, making it crucial for detecting contamination in cell culture laboratories.
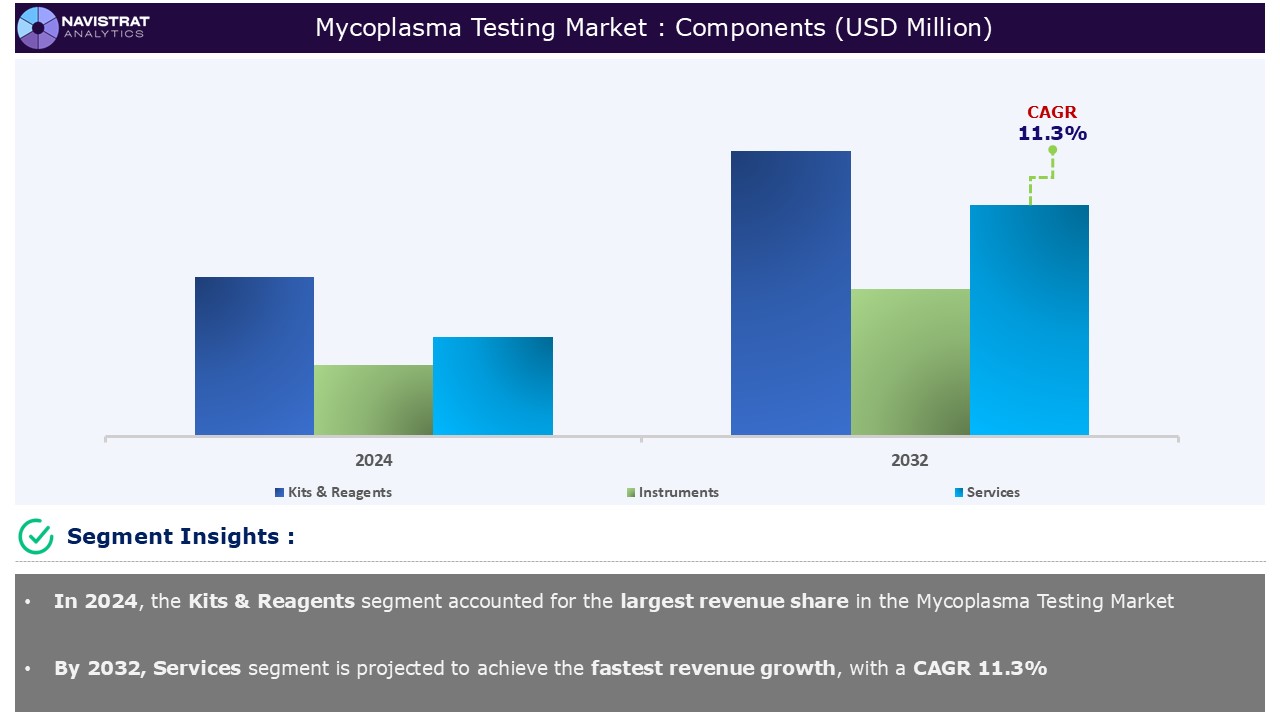
Application Segment Insights and Analysis:
Based on the Application, the Mycoplasma Testing Market is segmented into Cell Line Testing, Virus Testing, Biopharmaceutical Manufacturing, Vaccine Production, Gene Therapy and Cell Therapy, Stem Cell Research, and Others.
The Vaccine Production segment is expected to register significant revenue growth by 2032. The FDA has issued guidance to manufacturers of viral vaccines on the characterization and qualification of cell substrates, viral seeds, and other biological materials used in vaccine production, categorizing them as primary, diploid, or continuous. The Frederick National Laboratory for Cancer Research (FNLR) is developing a biopharmaceutical program for bacterial and yeast cells.
The project involves testing plasmid DNA or chromosomally localized exogenous genes, including DNA sequencing, restriction mapping, and strain typing. Also, the project requires determining plasmid copy number, restriction mapping, and determining the integrity of the construct in End of Production Cells (EOP). Outsourced tests will be performed by BQA-qualified vendors. The project accepts correct cell banks and adheres to expected fragment sizes.
End-Use Segment Insights and Analysis:
Based on the End-Use, the Mycoplasma Testing Market is segmented into Pharmaceuticals and Biotechnology, Contract Research Organizations (CROs), Contract Development and Manufacturing Organizations (CDMOs), Academic & Research Institutes, Hospitals & Clinics, and Veterinary.
The pharmaceuticals and Biotechnology segment contributed a significant revenue share in 2024. This segment is expected to experience significant growth in the historical period owing to the importance of mycoplasma testing in preventing batch failures caused by microbial contamination.
Early detection of Mycoplasma, a notorious contaminant in cell cultures, is vital for maintaining the integrity of biologics like vaccines and monoclonal antibodies, minimizing production losses, and ensuring compliance with stringent U.S. FDA standards. Mycoplasma testing is crucial for identifying microbes early in production, reducing losses. Government support and increased Research and Development activities are expected to drive market growth in this segment.
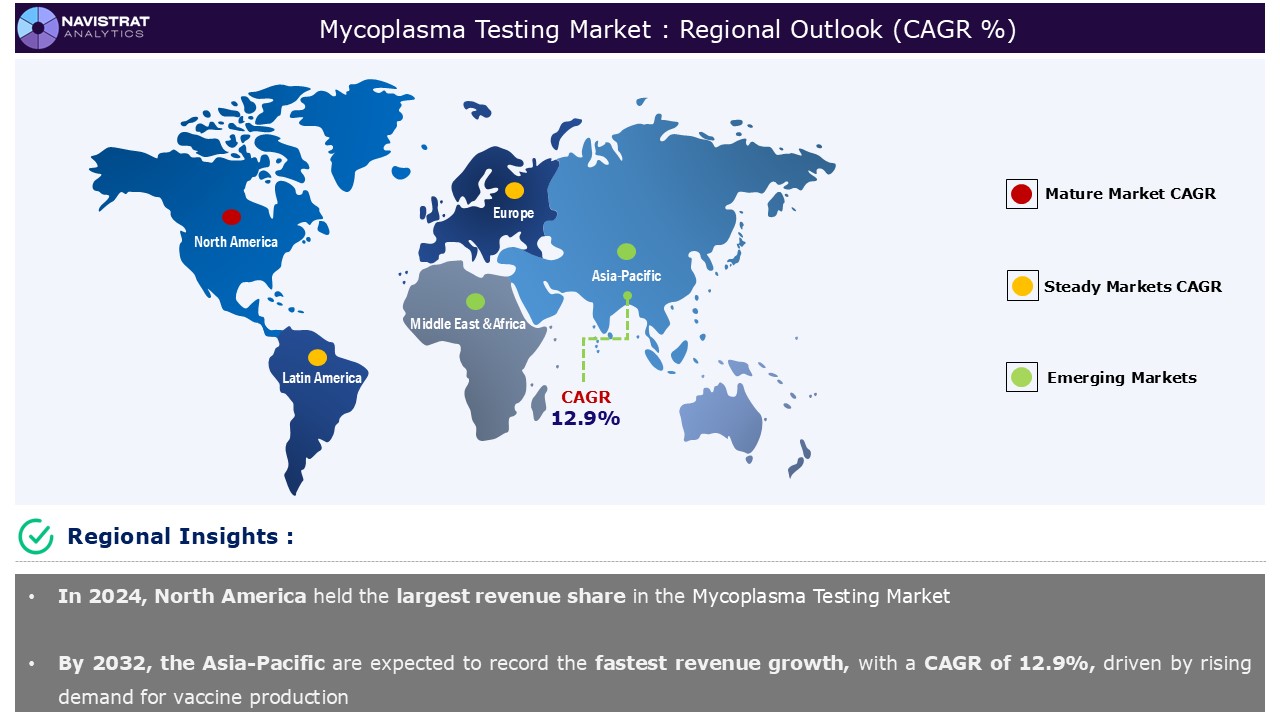
Geographical Outlook:
The Mycoplasma Testing Market is strategically segmented by geography to provide a comprehensive understanding of regional market dynamics. Discover demand analysis, emerging trends, and growth opportunities shaping market performance across different regions and countries.
North America Mycoplasma Testing Market:
North America registered the highest market revenue share in the mycoplasma testing market. The market in this region is driven by the increasing number of life science researchers and the presence of clinical and pre-clinical research companies such as Charles River Laboratories, IDEXX Laboratories, Thermo Fisher Scientific, and end-users like biotechnology, biological, and pharmaceutical companies.
In the United States, mycoplasma infections are common among populations. According to the U.S. Centers for Disease Control and Prevention (CDC), M. pneumoniae infections are widespread in the United States, with an estimated 2 million infections annually. The disease peaks every 3-7 years, with summer and early fall being more common. Outbreaks occur in crowded environments, with long incubation periods extending the duration. Stringent FDA regulations mandate mycoplasma testing for biologics, which are further driving adoption among pharmaceutical companies and CROs.
Asia Pacific Mycoplasma Testing Market:
Asia Pacific is expected to register the fastest revenue growth, reaching 12.9% by 2032. The increasing prevalence of cancer diseases and the growth in testing and trials are projected to fuel revenue growth in this region. According to the World Health Organization (WHO), every year, more than 2.3 million individuals in South East Asia are diagnosed with cancer, and around 1.4 million dies as a result of the disease.
Also, Increased demand for better laboratory and clinical research is significantly supporting to expansion of market share. In the first quarter of 2025, infections such as COVID-19, Human metapneumovirus (HMPV) have witnessed a major spike in several countries in Asia Pacific. As a result, the demand for testing kits and instruments is projected to grow steadily in 2025. Furthermore, regulatory rules implemented by healthcare organizations in favor of improving biosafety quality are expected to boost future growth potential.
Europe Mycoplasma Testing Market:
Europe contributed a substantial revenue share in 2024. Rising demand for mycoplasma testing for biosafety testing, analytical development, and cell bank are propelling the growth the market in this region. Companies across Europe are investing significantly in drug development and commercialization.
For instance, on 11th June 2024, Europe-based prominent contract design and manufacturing organization (CDMO), Freudenberg Medical, is strengthening its presence in the drug-device combination product market with a USD 50 million investment in Aachen, Germany. Moreover, on 14th March 2025, Johnson & Johnson is investing USD 150 million to expand their Ghent production facility in Belgium.
Competition Analysis:
The Mycoplasma Testing Market is characterized by numerous players, with major players competing across segments and regions. The list of major players included in the Mycoplasma Testing Market report is:
- Thermo Fisher Scientific Inc. (U.S.)
- Merck KGaA (Germany)
- InvivoGen (France)
- Lonza Group AG (Switzerland)
- Charles River Laboratories (U.S.)
- Eurofins Genomics Shared Services GmbH (Germany)
- Hoffmann-La Roche Ltd (Switzerland)
- IDEXX Laboratories, Inc. (U.S.)
- Nelson Laboratories, LLC (U.S.)
- Bionique Testing Laboratories LLC (U.S.)
- Krishgen Biosystems (U.S.)
- BaseClear BV (Netherlands)
- Biomérieux S.A. (France)
- Takara Bio Inc. (Japan)
- Generi Biotech (Czech Republic)
Strategic Developments in the Mycoplasma Testing Market:
- On 29th May 2025, Bionique Testing Laboratories entered into a strategic partnership with Cellipont Bioservices, a leading CDMO focused on cell therapy solutions. This collaboration is aimed at promoting the implementation and validation of rapid mycoplasma detection methods to support current Good Manufacturing Practices (cGMP) in the production of cell and gene therapies. Bionique will contribute its expertise by offering advanced mycoplasma testing services and customized validation support for specific products.
- On 9th November 2023, QIAGEN N.V. introduced QIAcuity Mycoplasma Quant Kit. This kit will help researchers to reliably detect Mycoplasma contamination throughout the research and production processes of cell and gene therapies. This kit features a highly sensitive, streamlined workflow that has undergone independent third-party validation and aligns with regulatory standards set by the U.S., European, and Japanese Pharmacopeias.
Key Advantages for Stakeholders:
Navistrat Analytics’ industry report provides an in-depth quantitative analysis of various market segments, historical and current trends, market forecasts, and dynamics within the global market. The historical years covered in this report are 2022 to 2023, with 2024 serving as the base year for market size calculations. The forecast period extends from 2025 to 2032.
The report includes an executive summary and a comprehensive overview of market drivers, restraints, opportunities, and challenges (DROC), along with insights into regulatory standards. It features detailed analyses such as PORTER’s Five Forces, SWOT, and PESTLE, as well as assessments of technological trends and the competitive landscape.
PORTER’s Five Forces analysis helps stakeholders evaluate the impact of new entrants, competitive rivalry, supplier power, buyer power, and substitution threats, enabling them to assess the level of competition and the attractiveness of the global market. The competitive landscape provides stakeholders with a clear understanding of the current market positions of key players, offering valuable insights into their competitive environment.
Scope And Key Highlights of The Mycoplasma Testing Market Report:
| Report Features | Details |
| Market Size in 2024 | USD 886.6 Million |
| Market Growth Rate in CAGR (2025–2032) | 9.3% |
| Market Revenue forecast to 2032 | USD 1,783.7 Million |
| Base year | 2024 |
| Historical year | 2022–2023 |
| Forecast period | 2025–2032 |
| Report Pages | 450 |
| Segments covered |
|
| Regional scope |
|
| Country Scope |
|
| Key Market Players |
|
| Delivery Format | Reports are delivered in PDF format via email. |
| Customization scope | Request for Customization |
The Mycoplasma Testing Market report offers a detailed analysis of market size, including historical revenue (in USD Million) data for 2022-2023 and revenue forecasts for 2025-2032 across the following segments:
- Components Outlook (Revenue, USD Million; 2022-2032)
- Kits & Reagents
- Polymerase chain reaction (PCR) Assays
- Nucleic Acid Detection Kits
- ELISA (Enzyme-Linked Immunosorbent Assay) Kits
- Staining
- Others
- Instruments
- PCR Systems
- qPCR Systems
- DNA Sequencers
- Others
- Services
- Testing Services
- Certification & Validation Services
- Consulting Services
- Kits & Reagents
- Testing Technology Outlook (Revenue, USD Million; 2022-2032)
- PCR-based
- ELISA-based
- Direct Assay
- Indirect Assay
- Next-Generation Sequencing (NGS)
- Others
- Application Outlook (Revenue, USD Million; 2022-2032)
- Cell Line Testing
- Virus Testing
- Biopharmaceutical Manufacturing
- Vaccine Production
- Gene Therapy and Cell Therapy
- Stem Cell Research
- Others
- End-Use Outlook (Revenue, USD Million; 2022-2032)
- Pharmaceuticals and Biotechnology
- Contract Research Organizations (CROs)
- Contract Development and Manufacturing Organizations (CDMOs)
- Academic & Research Institutes
- Hospitals & Clinics
- Veterinary
- Regional Outlook (Revenue, USD Million; 2022-2032)
- North America
- U.S.
- Canada
- Mexico
- Europe
- Germany
- France
- U.K.
- Italy
- Spain
- Benelux
- Nordic Countries
- Rest of Europe
- Asia Pacific
- China
- India
- Japan
- South Korea
- Singapore
- Oceania
- ASEAN Countries
- Rest of APAC
- Latin America
- Brazil
- Rest of LATAM
- Middle East & Africa
- GCC Countries
- South Africa
- Israel
- Turkey
- Rest of MEA
- North America
Frequently Asked Questions (FAQ) About the Mycoplasma Testing Market Report
The market size of the mycoplasma testing market was 886.6 million in 2024.
The growth of the mycoplasma testing market is expected to register a compound annual growth rate (CAGR) of 9.3% over the forecast period.
Rising demand for cell and gene therapies is a major factor driving the revenue growth of the mycoplasma testing market.
The high cost of validated testing kits and systems is a major restraint on the mycoplasma testing market.
Asia Pacific accounts for the fastest revenue growth of 12.9%.
Kits & Reagents is the leading segment of the mycoplasma testing market in terms of components.
- Market Definition
- Research Objective
- Research Methodology
- Research Design
- Data Collection Methods
- Primary
- Secondary
- Market Size Estimation
- Top-down method
- Bottom-up method
- Forecasting Methodology
- Tools and Models Used
- Market Overview and Trends
- Market Size and Forecast
- Industry Analysis
- Market Driver, Restraints, Opportunity, and Challenges (DROC) Analysis
- Market Drivers
- Rising demand for cell and gene therapies
- Increasing adoption of single-use systems in bioprocessing
- Growth in monoclonal antibody and vaccine production
- Market Restraints
- High cost of validated testing kits and systems
- High initial investment for establishing testing infrastructure
- Market Opportunities
- Increase in pharmaceutical Research and Development (R&D) investments and activities
- Growing adoption of in-house testing over outsourcing
- Market Challenges
- Challenges in global regulatory frameworks
- Limited shelf life of PCR reagent kits
- Regulatory Landscape
- Overview of Single Use Bioreactors Regulation Framework
- North America (USA, Canada, Mexico)
- Europe (Germany, UK, France)
- Asia-Pacific (China, Japan, South Korea, India)
- Middle East & Africa (UAE, Israel, Saudi Arabia, South Africa)
- Latin America (Brazil)
- Strategic Insights
- Porter’s Five Forces Analysis
- PESTLE Analysis
- Price Trend Analysis
- Market/ Technology Trends of Mycoplasma Testing Market
- Integration of rapid qPCR with closed-loop single-use systems
- AI-enhanced image analytics for culture-based detection workflows
- Portable isothermal amplification devices for decentralized QC testing
- Integration of mycoplasma detection into PAT (Process Analytical Technology) frameworks
- Adoption of CRISPR-based biosensors for mycoplasma detection
- Recent Developments
- Funding and Investments
- Merger and Acquisition
- Expansion
- Partnership and Collaboration
- Product/ Service Launch
- Technological developments
- Overview of Single Use Bioreactors Regulation Framework
- Component Market Revenue Estimates and Forecasts, 2022-2032
- Kits & Reagents
- Polymerase chain reaction (PCR) Assays
- Nucleic Acid Detection Kits
- ELISA (Enzyme-Linked Immunosorbent Assay) Kits
- Staining
- Others
- Instruments
- PCR Systems
- qPCR Systems
- DNA Sequencers
- Others
- Services
- Testing Services
- Certification & Validation Services
- Consulting Services
- Kits & Reagents
- Testing Technology Market Revenue Estimates and Forecasts, 2022-2032
- PCR-based Mycoplasma Detection
- ELISA-based Detection
- Direct Assay
- Indirect Assay
- Next-Generation Sequencing (NGS)
- Others
- Application Market Revenue Estimates and Forecasts, 2022-2032
- Cell Line Testing
- Virus Testing
- Biopharmaceutical Manufacturing
- Vaccine Production
- Gene Therapy and Cell Therapy
- Stem Cell Research
- Others
- End-Use Market Revenue Estimates and Forecasts, 2022-2032
- Pharmaceuticals and Biotechnology
- Contract Research Organizations (CROs)
- Contract Development and Manufacturing Organizations (CDMOs)
- Academic & Research Institutes
- Hospitals & Clinics
- Veterinary
- Mycoplasma Testing Market Revenue Estimates and Forecasts by Region, 2022-2032, USD Million
- North America
- North America Mycoplasma Testing Market by Component, Market Revenue Estimates and Forecasts, 2022-2032, USD Million
- Kits & Reagents
- Polymerase chain reaction (PCR) Assays
- Nucleic Acid Detection Kits
- ELISA (Enzyme-Linked Immunosorbent Assay) Kits
- Staining
- Others
- Instruments
- PCR Systems
- qPCR Systems
- DNA Sequencers
- Others
- Services
- Testing Services
- Certification & Validation Services
- Consulting Services
- Kits & Reagents
- North America Mycoplasma Testing Market by Testing Technology, Market Revenue Estimates and Forecasts, 2022-2032, USD Million
- PCR-based Mycoplasma Detection
- ELISA-based Detection
- Direct Assay
- Indirect Assay
- Next-Generation Sequencing (NGS)
- Others
- North America Mycoplasma Testing Market by Application, Market Revenue Estimates and Forecasts, 2022-2032, USD Million
- Cell Line Testing
- Virus Testing
- Biopharmaceutical Manufacturing
- Vaccine Production
- Gene Therapy and Cell Therapy
- Stem Cell Research
- Others
- North America Mycoplasma Testing Market by End-Use, Market Revenue Estimates and Forecasts, 2022-2032, USD Million
- Pharmaceuticals and Biotechnology
- Contract Research Organizations (CROs)
- Contract Development and Manufacturing Organizations (CDMOs)
- Academic & Research Institutes
- Hospitals & Clinics
- Veterinary
- North America Mycoplasma Testing Market Revenue Estimates and Forecasts by Country, 2022-2032, USD Million
- United States
- Canada
- Mexico
- North America Mycoplasma Testing Market by Component, Market Revenue Estimates and Forecasts, 2022-2032, USD Million
- Europe
- Europe Mycoplasma Testing Market by Component, Market Revenue Estimates and Forecasts, 2022-2032, USD Million
- Kits & Reagents
- Polymerase chain reaction (PCR) Assays
- Nucleic Acid Detection Kits
- ELISA (Enzyme-Linked Immunosorbent Assay) Kits
- Staining
- Others
- Instruments
- PCR Systems
- qPCR Systems
- DNA Sequencers
- Others
- Services
- Testing Services
- Certification & Validation Services
- Consulting Services
- Kits & Reagents
- Europe Mycoplasma Testing Market by Testing Technology, Market Revenue Estimates and Forecasts, 2022-2032, USD Million
- PCR-based Mycoplasma Detection
- ELISA-based Detection
- Direct Assay
- Indirect Assay
- Next-Generation Sequencing (NGS)
- Others
- Europe Mycoplasma Testing Market by Application, Market Revenue Estimates and Forecasts, 2022-2032, USD Million
- Cell Line Testing
- Virus Testing
- Biopharmaceutical Manufacturing
- Vaccine Production
- Gene Therapy and Cell Therapy
- Stem Cell Research
- Others
- Europe Mycoplasma Testing Market by End-Use, Market Revenue Estimates and Forecasts, 2022-2032, USD Million
- Pharmaceuticals and Biotechnology
- Contract Research Organizations (CROs)
- Contract Development and Manufacturing Organizations (CDMOs)
- Academic & Research Institutes
- Hospitals & Clinics
- Veterinary
- Europe Mycoplasma Testing Market Revenue Estimates and Forecasts by Country, 2022-2032, USD Million
- Germany
- United Kingdom
- France
- Italy
- Spain
- Benelux
- Nordic Countries
- Europe Mycoplasma Testing Market by Component, Market Revenue Estimates and Forecasts, 2022-2032, USD Million
- Asia-Pacific
- Asia-Pacific Mycoplasma Testing Market by Component, Market Revenue Estimates and Forecasts, 2022-2032, USD Million
- Kits & Reagents
- Polymerase chain reaction (PCR) Assays
- Nucleic Acid Detection Kits
- ELISA (Enzyme-Linked Immunosorbent Assay) Kits
- Staining
- Others
- Instruments
- PCR Systems
- qPCR Systems
- DNA Sequencers
- Others
- Services
- Testing Services
- Certification & Validation Services
- Consulting Services
- Kits & Reagents
- Asia-Pacific Mycoplasma Testing Market by Testing Technology, Market Revenue Estimates and Forecasts, 2022-2032, USD Million
- PCR-based Mycoplasma Detection
- ELISA-based Detection
- Direct Assay
- Indirect Assay
- Next-Generation Sequencing (NGS)
- Others
- Asia-Pacific Mycoplasma Testing Market by Application, Market Revenue Estimates and Forecasts, 2022-2032, USD Million
- Cell Line Testing
- Virus Testing
- Biopharmaceutical Manufacturing
- Vaccine Production
- Gene Therapy and Cell Therapy
- Stem Cell Research
- Others
- Asia-Pacific Mycoplasma Testing Market by End-Use, Market Revenue Estimates and Forecasts, 2022-2032, USD Million
- Pharmaceuticals and Biotechnology
- Contract Research Organizations (CROs)
- Contract Development and Manufacturing Organizations (CDMOs)
- Academic & Research Institutes
- Hospitals & Clinics
- Veterinary
- Asia-Pacific Mycoplasma Testing Market Revenue Estimates and Forecasts by Country, 2022-2032, USD Million
- China
- India
- Japan
- South Korea
- Singapore
- Oceania
- ASEAN Countries
- Rest of Asia-Pacific
- Asia-Pacific Mycoplasma Testing Market by Component, Market Revenue Estimates and Forecasts, 2022-2032, USD Million
- Latin America
- Latin America Mycoplasma Testing Market by Component, Market Revenue Estimates and Forecasts, 2022-2032, USD Million
- Kits & Reagents
- Polymerase chain reaction (PCR) Assays
- Nucleic Acid Detection Kits
- ELISA (Enzyme-Linked Immunosorbent Assay) Kits
- Staining
- Others
- Instruments
- PCR Systems
- qPCR Systems
- DNA Sequencers
- Others
- Services
- Testing Services
- Certification & Validation Services
- Consulting Services
- Kits & Reagents
- Latin America Mycoplasma Testing Market by Testing Technology, Market Revenue Estimates and Forecasts, 2022-2032, USD Million
- PCR-based Mycoplasma Detection
- ELISA-based Detection
- Direct Assay
- Indirect Assay
- Next-Generation Sequencing (NGS)
- Others
- Latin America Mycoplasma Testing Market by Application, Market Revenue Estimates and Forecasts, 2022-2032, USD Million
- Cell Line Testing
- Virus Testing
- Biopharmaceutical Manufacturing
- Vaccine Production
- Gene Therapy and Cell Therapy
- Stem Cell Research
- Others
- Latin America Mycoplasma Testing Market by End-Use, Market Revenue Estimates and Forecasts, 2022-2032, USD Million
- Pharmaceuticals and Biotechnology
- Contract Research Organizations (CROs)
- Contract Development and Manufacturing Organizations (CDMOs)
- Academic & Research Institutes
- Hospitals & Clinics
- Veterinary
- Latin America Mycoplasma Testing Market Revenue Estimates and Forecasts by Country, 2022-2032, USD Million
- Brazil
- Rest of LATAM
- Latin America Mycoplasma Testing Market by Component, Market Revenue Estimates and Forecasts, 2022-2032, USD Million
- Middle East & Africa
- Middle East & Africa Mycoplasma Testing Market by Component, Market Revenue Estimates and Forecasts, 2022-2032, USD Million
- Kits & Reagents
- Polymerase chain reaction (PCR) Assays
- Nucleic Acid Detection Kits
- ELISA (Enzyme-Linked Immunosorbent Assay) Kits
- Staining
- Others
- Instruments
- PCR Systems
- qPCR Systems
- DNA Sequencers
- Others
- Services
- Testing Services
- Certification & Validation Services
- Consulting Services
- Kits & Reagents
- Middle East & Africa Mycoplasma Testing Market by Testing Technology, Market Revenue Estimates and Forecasts, 2022-2032, USD Million
- PCR-based Mycoplasma Detection
- ELISA-based Detection
- Direct Assay
- Indirect Assay
- Next-Generation Sequencing (NGS)
- Others
- Middle East & Africa Mycoplasma Testing Market by Application, Market Revenue Estimates and Forecasts, 2022-2032, USD Million
- Cell Line Testing
- Virus Testing
- Biopharmaceutical Manufacturing
- Vaccine Production
- Gene Therapy and Cell Therapy
- Stem Cell Research
- Others
- Middle East & Africa Mycoplasma Testing Market by End-Use, Market Revenue Estimates and Forecasts, 2022-2032, USD Million
- Pharmaceuticals and Biotechnology
- Contract Research Organizations (CROs)
- Contract Development and Manufacturing Organizations (CDMOs)
- Academic & Research Institutes
- Hospitals & Clinics
- Veterinary
- Middle East & Africa Mycoplasma Testing Market Revenue Estimates and Forecasts by Country, 2022-2032, USD Million
- GCC Countries
- South Africa
- Israel
- Turkey
- Rest of Middle East & Africa
- Middle East & Africa Mycoplasma Testing Market by Component, Market Revenue Estimates and Forecasts, 2022-2032, USD Million
- North America
- Market Share Analysis
- Revenue Market Share by Key Players (2023-2024)
- Analysis of Top Players by Market Presence
- Competitive Matrix
- Competitive Strategies
- Mergers and Acquisitions
- Partnerships and Collaboration
- Investment and Fundings
- Agreement
- Expansion
- New Product/ Services Launches
- Technological Innovations
- Thermo Fisher Scientific Inc.
- Company Overview
- Financial Insights
- Product/ Services Offerings
- Strategic Developments
- SWOT Analysis
- Merck KGaA
- Company Overview
- Financial Insights
- Product/ Services Offerings
- Strategic Developments
- SWOT Analysis
- InvivoGen
- Company Overview
- Financial Insights
- Product/ Services Offerings
- Strategic Developments
- SWOT Analysis
- Lonza Group AG
- Company Overview
- Financial Insights
- Product/ Services Offerings
- Strategic Developments
- SWOT Analysis
- Charles River Laboratories International, Inc.
- Company Overview
- Financial Insights
- Product/ Services Offerings
- Strategic Developments
- SWOT Analysis
- Eurofins Genomics Shared Services GmbH
- Company Overview
- Financial Insights
- Product/ Services Offerings
- Strategic Developments
- SWOT Analysis
- Hoffmann-La Roche Ltd
- Company Overview
- Financial Insights
- Product/ Services Offerings
- Strategic Developments
- SWOT Analysis
- IDEXX Laboratories, Inc.
- Company Overview
- Financial Insights
- Product/ Services Offerings
- Strategic Developments
- SWOT Analysis
- Nelson Laboratories, LLC
- Company Overview
- Financial Insights
- Product/ Services Offerings
- Strategic Developments
- SWOT Analysis
- Bionique Testing Laboratories LLC
- Company Overview
- Financial Insights
- Product/ Services Offerings
- Strategic Developments
- Krishgen Biosystems
- Company Overview
- Financial Insights
- Product/ Services Offerings
- Strategic Developments
- SWOT Analysis
- BaseClear BV
- Company Overview
- Financial Insights
- Product/ Services Offerings
- Strategic Developments
- SWOT Analysis
- Biomérieux S.A.
- Company Overview
- Financial Insights
- Product/ Services Offerings
- Strategic Developments
- SWOT Analysis
- Takara Bio Inc.
- Company Overview
- Financial Insights
- Product/ Services Offerings
- Strategic Developments
- SWOT Analysis
- Generi Biotech
- Company Overview
- Financial Insights
- Product/ Services Offerings
- Strategic Developments
- SWOT Analysis
- Hologic, Inc.
- Company Overview
- Financial Insights
- Product/ Services Offerings
- Strategic Developments
- SWOT Analysis
- Qiagen N.V.
- Company Overview
- Financial Insights
- Product/ Services Offerings
- Strategic Developments
- SWOT Analysis

