CAR T-Cell Therapy Market Overview and Key Insights:
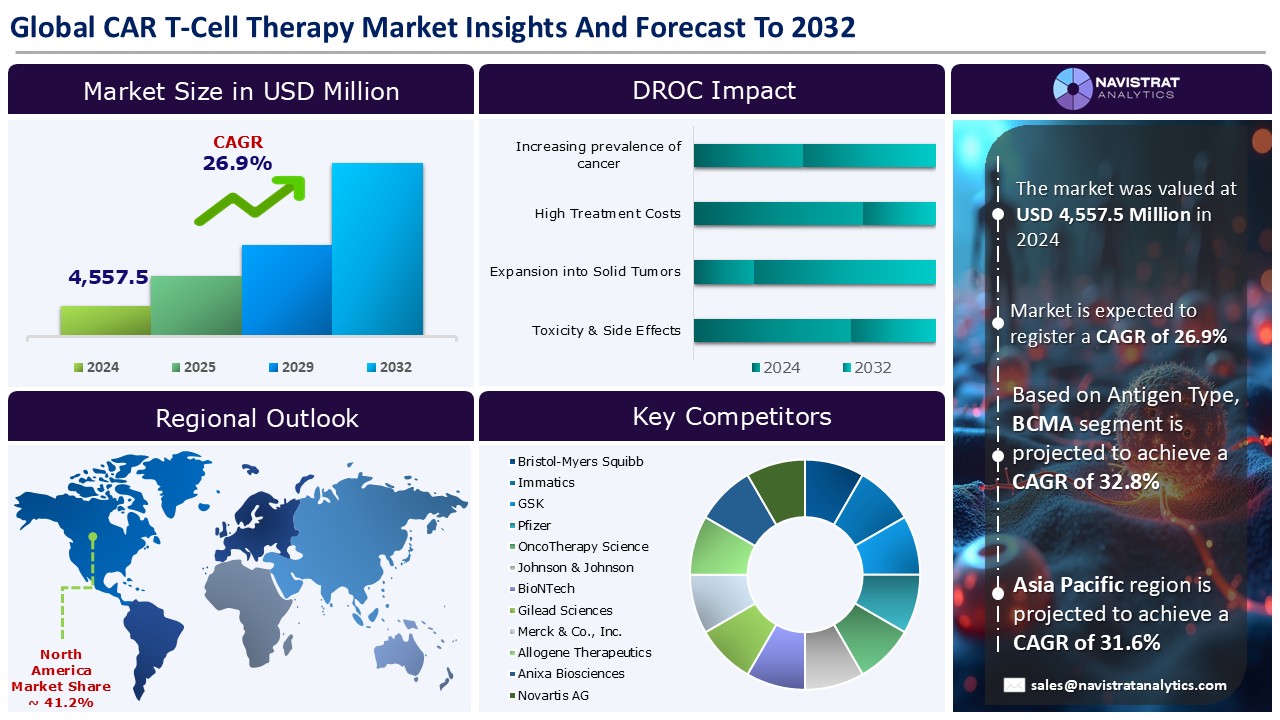
The CAR T-cell therapy market size reached USD 4,557.5 million in 2024 and is expected to register a revenue CAGR of 26.9% during the forecast period. CAR T-cell therapy is a type of cancer immunotherapy treatment that employs genetically modified immune cells known as T cells to better locate and destroy cancer cells.
Market Drivers:
Increase in prevalence of cancer is a key driver of revenue growth in the CAR T-cell therapy market. According to the American Cancer Society, leukemia, lymphoma, or myeloma diagnoses occur in one person in the US approximately every 3 minutes. In 2024, experts estimate that leukemia, lymphoma, or myeloma will affect a combined total of 187,740 people in the US. These new cases will account for 9.4 percent of the estimated 2,001,140 new cancer diagnoses expected in the US in 2024.
Technological advancements in CAR T-cell therapy is another key driver driving the market growth over the forecasted period. CAR-T cell therapy has transformed the treatment landscape for lymphoma, leukemia, and multiple myeloma, and these advances may soon apply to solid tumors, as well as autoimmune disorders and diseases such as type 1 diabetes and HIV, which have previously resisted effective treatment. Manufacturing innovations, such as automated processes and off-the-shelf CAR-T cells, will increase treatment accessibility and cost-effectiveness. Cardiac and liver fibrosis are currently being investigated for CAR-T cell treatment following the development of a distinct endogenous marker for cardiac fibroblasts.
On January 2025, Umoja Biopharma has raised USD 100 million in Series C funding as the cancer-focused biotech seeks to advance more of its cell therapies into clinical trials. Umoja’s primary clinical-stage candidate is UB-TT170, a TumorTag-based therapy that targets solid tumor cells expressing folate receptor alpha. The asset is being tested in a phase 1 trial alongside a CAR-T therapy for osteosarcoma, the most frequent kind of bone malignancy in children.
Market Opportunity:
Expansion into solid tumors acts as an opportunity for the CAR T-cell therapy market. Solid tumors, unlike blood malignancies, exist in an immune-suppressing microenvironment. Furthermore, dense clusters of malignant cells may form a barrier, preventing CARs from delivering their cancer-fighting capabilities into cells. The lack of targets with restricted expression on tumor cell surfaces is a significant constraint in the development of CAR-T treatments for solid tumors. CAR-T cell treatment for solid tumors, which targets multiple cell surface antigens, has shown some promise in pre-clinical development and clinical studies.
On August 2024, the FDA has given accelerated approval to afamitresgene autoleucel (TECELRA, Adaptimmune, LLC), a genetically modified autologous T cell immunotherapy directed by melanoma-associated antigen A4 (MAGE-A4), for adults with metastatic or unresectable synovial sarcoma who have undergone chemotherapy, are HLA-A*02:01P, -A*02:02P, -A*02:03P, or -A*02:06P, and whose tumor expresses the MAGE-A4 antigen as indicated by FDA-approved or cleared companion diagnostic devices.
Recent Trends:
Emerging trends include expansion into autoimmune diseases, growth of allogeneic CAR-T therapies, integration of digital health and AI, and new target antigens and combination therapies.
Experts expect regulatory approval of CAR T-cell treatment as a therapeutic option for severe refractory autoimmune illnesses in the coming years. Similarly, researchers are exploring CAR T-cell therapy as a potential treatment for various non-malignant disorders, including fibrosis, based on compelling preclinical findings. Scientists have successfully used CAR T cells to treat autoimmune disorders, achieving a more thorough depletion of tissue-resident B cells than monoclonal antibodies. As a living drug, CAR-T cells can achieve sustained depletion without the need for frequent administration, holding the promise for durable remissions in various autoimmune diseases, which is a significant advancement over conventional therapies.
Restraints & Challenges:
High treatment costs is a key restraint limiting the growth of the market. Chimeric antigen receptor (CAR) T-cells are a cellular immunotherapy with amazing efficiency in treating a variety of hematologic malignancies, however they come at exorbitant pricing, making them prohibitively expensive in many countries. Drug prices in the United States and around the world have risen dramatically over the last two decades, with the median launch price for cancer treatments in the US currently exceeding USD 155,000 per year. Cellular and gene therapies are some of the most expensive treatments on the market.
Antigen Type Segment Insights and Analysis:
Based on the antigen type, the CAR T-cell therapy market is segmented into CD19, BCMA, CD22, CD20, and others.
CD19 segment contributed the largest market share in 2024. B cells express CD19, a surface protein present throughout their development, which is why it appears in nearly all B-cell malignancies, including chronic lymphocytic leukemia (CLL), acute lymphoblastic leukemia (ALL), and many non-Hodgkin lymphomas. Researchers consider CD19 a promising target for CAR-modified T-cell treatments because it is expressed almost universally and specifically in a single cell lineage. Scientists are currently investigating CD19-directed CAR-modified T-cell treatments for B-cell malignancies as the most advanced form of engineered T-cell therapies. Clinical trials in CLL and ALL have demonstrated strong activity and produced impressive clinical outcomes.
On March 2024, Bristol Myers Squibb announced that the U.S. Food and Drug Administration (FDA) has granted accelerated approval for Breyanzi (lisocabtagene maraleucel; liso-cel), a CD19-targeting chimeric antigen receptor (CAR) T cell therapy. This approval covers the treatment of adult patients with relapsed or refractory chronic lymphocytic leukemia (CLL) or small lymphocytic lymphoma (SLL) who have undergone at least two previous treatments, including a Bruton tyrosine kinase (BTK) inhibitor and a B-cell lymphoma 2 (BCL-2) inhibitor.
The BCMA segment is projected to grow at the fastest rate throughout the forecast period. Researchers have targeted B cell maturation antigen (BCMA)—which is selectively expressed in both malignant and normal plasma cells—using numerous immunotherapeutic approaches. Chimeric antigen receptor (CAR) T cells have transformed the treatment of B cell malignancies and significantly improved the prognosis of relapsed/refractory multiple myeloma (RRMM), marking a breakthrough in cancer immunotherapy. The most advanced CAR T cell therapy for MM is BCMA-targeting CAR T cell therapy; idecabtagene vicleucel (Ide-cel) and ciltacabtagene autoleucel (Cilta-cel) have already received approval from the US Food and Drug Administration for MM.
On March 2024, CARsgen Therapeutics Holdings Limited, a company specializing in innovative CAR T-cell therapies for hematologic malignancies and solid tumors, has announced that China’s National Medical Products Administration has approved the New Drug Application for zevorcabtagene autoleucel. This autologous CAR-T therapy targets BCMA and is intended for adult patients with relapsed or refractory multiple myeloma who have undergone at least three prior lines of treatment, including a proteasome inhibitor and an immunomodulatory agent.

Indication Segment Insights and Analysis:
Based on the indication, the CAR T-Cell Therapy market is segmented into leukemia, lymphoma, multiple myleoma, autoimmune disorders, and others.
Lymphoma segment contributed the largest market share in 2024. According to the American Cancer Society, Non-Hodgkin lymphoma (NHL) is among the most frequent tumors in the United States, accounting for around 4% of all cancers. Approximately 80,350 persons (45,140 men and 35,210 women) will be diagnosed with NHL. This encompasses both adults and children. This malignancy will kill approximately 19,390 persons (11,060 men and 8,330 women).
On January 2025, March Biosciences, an emerging clinical stage biotechnology company dedicated to combating difficult cancers and other diseases, announced that the United States Food and Drug Administration (FDA) has granted orphan drug designation to MB-105, the company’s first-in-class CD5-targeted CAR-T cell therapy, for the treatment of relapsed / refractory CD5-positive T-cell lymphoma.
Autoimmune disorders are expected to have the highest market growth rate throughout the forecasted period. According to the Centre of Disease Control and Prevention (CDC), Lupus erythematosus (SLE) is the most frequent kind of lupus. Over 200,000 persons in the United States are believed to have SLE. Most persons with lupus are women. It is estimated that 9 out of 10 persons with lupus are female. Women of childbearing age (15 to 44 years) are more likely to acquire SLE. Black or African American, Hispanic, Asian, and American Indian and Alaska Native (AI/AN) groups are more vulnerable than White people.
In January 2025, the University of Chicago Medicine began Phase 2 clinical studies to investigate how CAR T-cell therapy can treat three autoimmune diseases: systemic lupus erythematosus, inflammatory myositis, and systemic sclerosis. Cabaletta, a company specializing in developing CAR T cells for rheumatologic conditions, is supporting the trials, which take place at nine sites, including UChicago Medicine.
End-Use Segment Insights and Analysis:
Based on the end-use, the CAR T-cell therapy market is segmented into hospitals & cancer treatment centers, academic & research institutes, and pharmaceutical & biotechnology companies.
Pharmaceutical & biotechnology companies segment contributed the largest market share in 2024. The pharmaceutical sector remains a hotspot of innovation, driven by the evolution of new treatment paradigms, the gravity of unmet need, and the increasing importance of technologies such as pharmacogenomics, digital medicines, and artificial intelligence. In recent years, novel cell types and technologies, including as CAR-T (Chimeric Antigen Receptor T-cell) therapies, have emerged as a transformative option to address the hurdles given by current treatments and the nature of targeted diseases.
On November 2023, Legend Biotech Corporation, a global biotechnology company focused on developing, manufacturing, and commercializing innovative therapies for life-threatening diseases, announced that its wholly owned subsidiary, Legend Biotech Ireland Limited, has entered into an exclusive worldwide licensing agreement with Novartis Pharma AG. This agreement covers certain Legend Biotech chimeric antigen receptor T-cell (CAR-T) therapies targeting DLL3, including the autologous CAR-T candidate LB2102. Under the agreement, Novartis gains exclusive global rights to develop, produce, and market these therapies, with the potential to incorporate its T-Charge platform into their manufacturing process.
Hospitals & cancer treatment centers segment is expected to have the highest growth rate throughout the forecasted period. Hospitals have made significant investments in cutting-edge infrastructure, put together groups of highly qualified medical professionals and researchers, and carried out in-depth studies and clinical trials. A significant advancement in cancer treatment, CAR T-cell therapy gives patients who have run out of conventional treatments new hope. This novel method uses the body’s immune system to specifically target and destroy cancer cells, which may result in long-lasting remissions and enhanced quality of life. Clinical trials investigating CAR T-cell therapy’s ability to treat solid tumors and other cancer types are part of ongoing research to broaden its applications. In the battle against cancer, this individualized and focused strategy is a potential new frontier that gives patients and their families fresh hope and opportunities.
Geographical Outlook:
CAR T-cell therapy market is strategically segmented by geography to provide a comprehensive understanding of regional market dynamic. Discover demand analysis, emerging trends, and growth opportunities shaping market performance across different region and countries.
North America CAR T-Cell Therapy Market:
North America is registered to have the highest market share in CAR T-cell therapy market in 2024. This is mainly due to the rising prevalence of cancer and a rising demand for personalized medicine. According to the American Cancer Institute, experts estimate that 36,110 new cases of multiple myeloma will be diagnosed in 2025 (20,030 in men and 16,080 in women). They anticipate that 12,030 people will die from the disease (6,540 men and 5,490 women). In the United States, individuals face a lifetime risk of developing multiple myeloma of less than 1%—approximately 1 in 108 for men and 1 in 133 for women. Elderly adults develop most cases of multiple myeloma, while people under the age of 35 account for less than 1% of diagnoses. Most patients with multiple myeloma are 65 years of age or older.
On May 2024, Poseida Therapeutics, Inc., and Astellas Pharma Inc. announced that Xyphos Biosciences, Inc. and Poseida have signed a license agreement and research collaboration to combine their respective cutting-edge cell therapy platforms to develop unique convertibleCAR initiatives. Allogeneic CAR-T cell therapy product candidates for solid and liquid malignancies are part of the oncology pipeline, which targets patient groups with significant unmet medical needs.
Asia Pacific CAR T-Cell Therapy Market:
Asia Pacific is registered the fastest growth rate during the forecasted period. The Asia-Pacific region has seen great advancements in precision medicine. Genomic sequencing, biomarker discovery, and the creation of extensive biobanks have advanced in countries like China, South Korea, and Singapore. Additionally, there is a growing focus on developing companion diagnostics to determine which patients will benefit most from drugs. According to estimates, 17.6% of women and 26.3% of men in Japan will get cancer by the age of 75.4. Against the backdrop of medical advancements and the high prevalence among individuals of working age, the number of survivors has been continuously rising, including for patients with breast and prostate cancer, two of the most frequent cancers.
On November 2024, Alloy Therapeutics Inc., a biotechnology ecosystem company committed to democratizing access to cutting-edge drug discovery technologies, announced a strategic collaboration and license agreement with Takeda Pharmaceutical Company Limited to develop Takeda’s proprietary induced pluripotent stem cell (iPSC)-derived CAR-T cell platform (iCAR-T) and iPSC-derived CAR-NK platform (iCAR-NK). Alloy will concentrate on speeding the development of critical treatments to combat solid and hematological cancers.
Europe CAR T-Cell Therapy Market:
Europe is to register a considerable market share during the forecasted period. This is mainly driven by growing preference of cancer and technological advancements in CAR T-cell therapy. Furthermore, approval and launch of new drugs is also augmenting the growth of the market in the region. For instance, on November 2023, Bristol Myers Squibb announced that the European Commission (EC) has approved Breyanzi (lisocabtagene maraleucel; liso-cel), a CD19-targeting chimeric antigen receptor (CAR) T cell therapy, for adult patients with diffuse large B-cell lymphoma (DLBCL), high-grade B-cell lymphoma (HGBCL), primary mediastinal large B-cell lymphoma (PMBCL), and follicular lymphoma grade 3B (FL3B). The approval applies to patients who have relapsed within 12 months after completing first-line chemoimmunotherapy or are refractory to it. This authorization extends to all European Union (EU) member states.

Competition Analysis:
The CAR T-cell therapy market is characterized by a fragmented structure, with several players competing across various segments and regions. List of major players included in the CAR T-cell therapy market report are:
- Bristol-Myers Squibb
- Immatics
- GSK
- Pfizer
- OncoTherapy Science
- Johnson & Johnson
- BioNTech
- Gilead Sciences
- Merck & Co., Inc.
- Allogene Therapeutics
- Anixa Biosciences
- Autolus Therapeutics
- Cartesian Therapeutics Inc
- Novartis AG
- CARsgen Therapeutics Holdings Limited
Strategic Developments in CAR T-Cell Therapy Market:
- In January 2025, Immuneel Therapeutics has launched Qartemi, India’s first global CAR T-cell therapy for adult B-cell non-Hodgkin lymphoma (BNHL). A tailored therapy for adult patients with relapsed or refractory B-NHL, the company, which is India’s foremost cell and gene therapy start-up, fills a vital vacuum in the country’s cancer immunotherapy scene. This therapy uses a patient’s immune cells to target and eradicate cancer, providing new hope to individuals fighting aggressive blood malignancies.
- In October 2024, AvenCell Therapeutics, Inc., a leading clinical-stage cell therapy business focusing on developing autologous and allogeneic switchable CAR-T cell therapeutics, has raised USD 112 million in Series B funding. The financing was spearheaded by Novo Holdings, a worldwide life sciences investor. The investment also included new investors F-Prime Capital, Eight Roads Ventures Japan, Piper Heartland Healthcare Capital, and NYBC Ventures, in addition to founding backer Blackstone Life Sciences.
- In September 2024, Arsenal Biosciences, Inc., a clinical-stage programmable cell therapy business focusing on developing breakthrough CAR T-cell therapeutics for solid malignancies, announced today the completion of an oversubscribed USD 325 million Series C funding transaction. The funding round included new investors ARCH Venture Partners, Milky Way Investments Group, Regeneron Ventures, NVentures (NVIDIA’s venture capital arm), Luma Group, funds and accounts advised by T. Rowe Price Associates, Inc., Rock Springs Capital, among others, with ongoing support from existing investors the Parker Institute for Cancer Immunotherapy (PICI), SoftBank Vision Fund 2, Bristol-Myers Squibb Company, Westlake Village BioPartners, Kleiner Perkins, Byers Capital, and Hitachi Ventures
Key Advantages for Stakeholders:
Navistrat Analytics’ industry report provides an in-depth quantitative analysis of various market segments, historical and current trends, market forecasts, and dynamics within the global market. The historical years covered in this report are 2022 to 2023, with 2024 serving as the base year for market size calculations. The forecast period extends from 2025 to 2032.
The report includes an executive summary and a comprehensive overview of market drivers, restraints, opportunities, and challenges (DROC), along with insights into regulatory standards. It features detailed analyses such as PORTER’s Five Forces, SWOT, and PESTLE, as well as assessments of technological trends and the competitive landscape.
PORTER’s Five Forces analysis helps stakeholders evaluate the impact of new entrants, competitive rivalry, supplier power, buyer power, and substitution threats, enabling them to assess the level of competition and the attractiveness of the global market. The competitive landscape provides stakeholders with a clear understanding of the current market positions of key players, offering valuable insights into their competitive environment.
Scope And Key Highlights Of The CAR T-Cell Therapy Market Report:
| Report Features | Details |
| Market Size in 2024 | USD 4,557.5 Million |
| Market Growth Rate in CAGR (2025–2032) | 26.9% |
| Market Revenue forecast to 2032 | USD 30,777.6 Million |
| Base year | 2024 |
| Historical year | 2022-2023 |
| Forecast period | 2025-2032 |
| Report Pages | 450 |
| Segments covered |
|
| Regional scope |
|
| Country Scope |
|
| Key Market Players |
|
| Delivery Format | Reports are delivered in PDF format via email. |
| Customization scope | Explore options (Forms) |
The CAR T-Cell Therapy market report offers a detailed analysis of market size, including historical revenue (in USD Million) data for 2022-2023 and revenue forecasts for 2025-2032 across the following segments:
- Type Outlook (Revenue, USD Million; 2022-2032)
- Kymriah
- Yescarta
- Tecartus
- Abecma
- Carvykti
- Others
- Sample Type Outlook (Revenue, USD Million; 2022-2032)
- CD19
- BCMA
- CD22
- CD20
- Others
- Therapy Type Outlook (Revenue, USD Million; 2022-2032)
- Autologous CAR T-cell Therapy
- Allogeneic CAR T-cell Therapy
- Indication Outlook (Revenue, USD Million; 2022-2032)
- Leukemia
- Lymphoma
- Multiple Myleoma
- Autoimmune Disorders
- Others
- End-Use Outlook (Revenue, USD Million; 2022-2032)
- Hospitals & Cancer Treatment Centers
- Academic & Research Institutes
- Pharmaceutical & Biotechnology Companies
- Regional Outlook (Revenue, USD Million; 2022-2032)
- North America
- U.S.
- Canada
- Mexico
- Europe
- Germany
- France
- U.K.
- Italy
- Spain
- Benelux
- Nordic Countries
- Rest of Europe
- Asia Pacific
- China
- India
- Japan
- South Korea
- Oceania
- ASEAN Countries
- Rest of APAC
- Latin America
- Brazil
- Rest of LATAM
- Middle East & Africa
- GCC Countries
- South Africa
- Israel
- Turkey
- Rest of MEA
- North America
Frequently Asked Questions (FAQ) about the CAR T-Cell Therapy market report
The market size of CAR T-cell therapy market was 4,557.5 million in 2024.
The market size of CAR T-Cell Therapy market is expected to register compound annual growth rate (CAGR) of 26.9% over the forecast period.
Increase in prevalence of cancer, technological advancements in Car T-cell therapy, and rising demand for personalized medicine are major key factors driving the market revenue growth of the CAR T-cell therapy market.
High treatment costs and complex manufacturing & supply chain issues are key limiting factors driving the market.
Asia Pacific account for fastest revenue growth of 31.6%.
CD19 is the major leading segment of CAR T-cell therapy market in terms of antigen type.
- Market Definition
- Research Objective
- Research Methodology
- Research Design
- Data Collection Applications
- Primary
- Secondary
- Market Size Estimation
- Top-down Application
- Bottom-up Application
- Forecasting Methodology
- Tools and Models Used
- Market Overview and Trends
- Market Size and Forecast
- Industry Analysis
- Market Driver, Restraints, Opportunity, and Challenges (DROC) Analysis
- Market Drivers
- Increase in prevalence of cancer
- Technological advancements in Car T-cell therapy
- Rising demand for personalized medicine
- Market Restraints
- High treatment costs
- Complex manufacturing & supply chain issues
- Market Opportunities
- Expansion into solid tumors
- Emergence of In Vivo CAR-T Therapy
- Combination therapies & next-generation CAR-Ts
- Market Challenges
- Toxicity & side effects
- Reimbursement issues
- Regulatory Landscape
- North America
- Europe
- Asia Pacific
- Latin America
- Middle East and Africa
- Strategic Insights
- Porter’s Five Forces Analysis
- PESTLE Analysis
- Price Trend Analysis
- Value Chain Analysis
- Technological Trends
- Recent Developments
- Funding
- Merger and Acquisition
- Expansion
- Partnership and Collaboration
- Product/ Service Launch
- Type Market Revenue Estimates and Forecasts, 2022-2032
- Kymriah
- Yescarta
- Tecartus
- Abecma
- Carvykti
- Others
- Antigen Type Market Revenue Estimates and Forecasts, 2022-2032
- CD19
- BCMA
- CD22
- CD20
- Others
- Therapy Type Market Revenue Estimates and Forecasts, 2022-2032
- Autologous CAR T-cell Therapy
- Allogeneic CAR T-cell Therapy
- Indication Market Revenue Estimates and Forecasts, 2022-2032
- Leukemia
- Lymphoma
- Multiple Myleoma
- Autoimmune Disorders
- Others
- End-Use Market Revenue Estimates and Forecasts, 2022-2032
- Hospitals & Cancer Treatment Centers
- Academic & Research Institutes
- Pharmaceutical & Biotechnology Companies
- CAR T-Cell Therapy Market Revenue Estimates and Forecasts by Region, 2022-2032, USD Million
- North America
- North America CAR T-Cell Therapy Market By Type, Market Revenue Estimates and Forecasts, 2022-2032, USD Million
- Kymriah
- Yescarta
- Tecartus
- Abecma
- Carvykti
- Others
- North America CAR T-Cell Therapy Market By Antigen Type, Market Revenue Estimates and Forecasts, 2022-2032, USD Million
- CD19
- BCMA
- CD22
- CD20
- Others
- North America CAR T-Cell Therapy Market By Therapy Type, Market Revenue Estimates and Forecasts, 2022-2032, USD Million
- Autologous CAR T-cell Therapy
- Allogeneic CAR T-cell Therapy
- North America CAR T-Cell Therapy Market By Indication, Market Revenue Estimates and Forecasts, 2022-2032, USD Million
- Leukemia
- Lymphoma
- Multiple Myleoma
- Autoimmune Disorders
- Others
- North America CAR T-Cell Therapy Market By End-Use, Market Revenue Estimates and Forecasts, 2022-2032, USD Million
- Hospitals & Cancer Treatment Centers
- Academic & Research Institutes
- Pharmaceutical & Biotechnology Companies
- North America CAR T-Cell Therapy Market Revenue Estimates and Forecasts by Country, 2022-2032, USD Million
- United States
- Canada
- Mexico
- North America CAR T-Cell Therapy Market By Type, Market Revenue Estimates and Forecasts, 2022-2032, USD Million
- North America
- Europe
- Europe CAR T-Cell Therapy Market By Type, Market Revenue Estimates and Forecasts, 2022-2032, USD Million
- Kymriah
- Yescarta
- Tecartus
- Abecma
- Carvykti
- Others
- Europe CAR T-Cell Therapy Market By Antigen Type, Market Revenue Estimates and Forecasts, 2022-2032, USD Million
- CD19
- BCMA
- CD22
- CD20
- Others
- Europe CAR T-Cell Therapy Market By Therapy Type, Market Revenue Estimates and Forecasts, 2022-2032, USD Million
- Autologous CAR T-cell Therapy
- Allogeneic CAR T-cell Therapy
- Europe CAR T-Cell Therapy Market By Indication, Market Revenue Estimates and Forecasts, 2022-2032, USD Million
- Leukemia
- Lymphoma
- Multiple Myleoma
- Autoimmune Disorders
- Others
- Europe CAR T-Cell Therapy Market By End-Use, Market Revenue Estimates and Forecasts, 2022-2032, USD Million
- Hospitals & Cancer Treatment Centers
- Academic & Research Institutes
- Pharmaceutical & Biotechnology Companies
- Europe CAR T-Cell Therapy Market Revenue Estimates and Forecasts by Country, 2022-2032, USD Million
- Germany
- United Kingdom
- France
- Italy
- Spain
- Benelux
- Nordic Countries
- Rest of Europe
- Europe CAR T-Cell Therapy Market By Type, Market Revenue Estimates and Forecasts, 2022-2032, USD Million
- Asia-Pacific
- Asia Pacific CAR T-Cell Therapy Market By Type, Market Revenue Estimates and Forecasts, 2022-2032, USD Million
- Kymriah
- Yescarta
- Tecartus
- Abecma
- Carvykti
- Others
- Asia Pacific CAR T-Cell Therapy Market By Antigen Type, Market Revenue Estimates and Forecasts, 2022-2032, USD Million
- CD19
- BCMA
- CD22
- CD20
- Others
- Asia Pacific CAR T-Cell Therapy Market By Therapy Type, Market Revenue Estimates and Forecasts, 2022-2032, USD Million
- Autologous CAR T-cell Therapy
- Allogeneic CAR T-cell Therapy
- Asia Pacific CAR T-Cell Therapy Market By Indication, Market Revenue Estimates and Forecasts, 2022-2032, USD Million
- Leukemia
- Lymphoma
- Multiple Myleoma
- Autoimmune Disorders
- Others
- Asia Pacific CAR T-Cell Therapy Market By End-Use, Market Revenue Estimates and Forecasts, 2022-2032, USD Million
- Hospitals & Cancer Treatment Centers
- Academic & Research Institutes
- Pharmaceutical & Biotechnology Companies
- Asia Pacific CAR T-Cell Therapy Market Revenue Estimates and Forecasts by Country, 2022-2032, USD Million
- China
- India
- Japan
- South Korea
- Oceania
- ASEAN Countries
- Rest of Asia-Pacific
- Asia Pacific CAR T-Cell Therapy Market By Type, Market Revenue Estimates and Forecasts, 2022-2032, USD Million
- Latin America
- Latin America CAR T-Cell Therapy Market By Type, Market Revenue Estimates and Forecasts, 2022-2032, USD Million
- Kymriah
- Yescarta
- Tecartus
- Abecma
- Carvykti
- Others
- Latin America CAR T-Cell Therapy Market By Antigen Type, Market Revenue Estimates and Forecasts, 2022-2032, USD Million
- CD19
- BCMA
- CD22
- CD20
- Others
- Latin America CAR T-Cell Therapy Market By Therapy Type, Market Revenue Estimates and Forecasts, 2022-2032, USD Million
- Autologous CAR T-cell Therapy
- Allogeneic CAR T-cell Therapy
- Latin America CAR T-Cell Therapy Market By Indication, Market Revenue Estimates and Forecasts, 2022-2032, USD Million
- Leukemia
- Lymphoma
- Multiple Myleoma
- Autoimmune Disorders
- Others
- Latin America CAR T-Cell Therapy Market By End-Use, Market Revenue Estimates and Forecasts, 2022-2032, USD Million
- Hospitals & Cancer Treatment Centers
- Academic & Research Institutes
- Pharmaceutical & Biotechnology Companies
- Latin America CAR T-Cell Therapy Market Revenue Estimates and Forecasts by Country, 2022-2032, USD Million
- Brazil
- Rest of Latin America
- Latin America CAR T-Cell Therapy Market By Type, Market Revenue Estimates and Forecasts, 2022-2032, USD Million
- Middle East & Africa
-
- Middle East & Africa CAR T-Cell Therapy Market By Type, Market Revenue Estimates and Forecasts, 2022-2032, USD Million
- Kymriah
- Yescarta
- Tecartus
- Abecma
- Carvykti
- Others
- Middle East & Africa CAR T-Cell Therapy Market By Antigen Type, Market Revenue Estimates and Forecasts, 2022-2032, USD Million
- CD19
- BCMA
- CD22
- CD20
- Others
- Middle East & Africa CAR T-Cell Therapy Market By Therapy Type, Market Revenue Estimates and Forecasts, 2022-2032, USD Million
- Autologous CAR T-cell Therapy
- Allogeneic CAR T-cell Therapy
- Middle East & Africa CAR T-Cell Therapy Market By Indication, Market Revenue Estimates and Forecasts, 2022-2032, USD Million
- Leukemia
- Lymphoma
- Multiple Myleoma
- Autoimmune Disorders
- Others
- Middle East & Africa CAR T-Cell Therapy Market By End-Use, Market Revenue Estimates and Forecasts, 2022-2032, USD Million
- Hospitals & Cancer Treatment Centers
- Academic & Research Institutes
- Pharmaceutical & Biotechnology Companies
- Middle East & Africa CAR T-Cell Therapy Market Revenue Estimates and Forecasts by Country, 2022-2032, USD Million
- GCC Countries
- South Africa
- Israel
- Turkey
- Rest of Middle East & Africa
- Middle East & Africa CAR T-Cell Therapy Market By Type, Market Revenue Estimates and Forecasts, 2022-2032, USD Million
- Market Share Analysis
- Revenue Market Share by Key Players (2023-2024)
- Analysis of Top Players by Market Presence
- Competitive Matrix
- Competitive Strategies
- Mergers and Acquisitions
- Partnerships and Collaboration
- Investment and Fundings
- Agreement
- Expansion
- New Product/ Services Launches
- Technological Innovations
- Bristol-Myers Squibb
- Company Overview
- Financial Insights
- Product/ Services Offerings
- Strategic Developments
- SWOT Analysis
- Immatics
- Company Overview
- Financial Insights
- Product/ Services Offerings
- Strategic Developments
- SWOT Analysis
- GSK
- Company Overview
- Financial Insights
- Product/ Services Offerings
- Strategic Developments
- SWOT Analysis
- Pfizer
- Company Overview
- Financial Insights
- Product/ Services Offerings
- Strategic Developments
- SWOT Analysis
- OncoTherapy Science
- Company Overview
- Financial Insights
- Product/ Services Offerings
- Strategic Developments
- SWOT Analysis
- Johnson & Johnson
- Company Overview
- Financial Insights
- Product/ Services Offerings
- Strategic Developments
- SWOT Analysis
- BioNTech
- Company Overview
- Financial Insights
- Product/ Services Offerings
- Strategic Developments
- SWOT Analysis
- Gilead Sciences
- Company Overview
- Financial Insights
- Product/ Services Offerings
- Strategic Developments
- SWOT Analysis
- Merck & Co., Inc.
- Company Overview
- Financial Insights
- Product/ Services Offerings
- Strategic Developments
- SWOT Analysis
- Allogene Therapeutics
- Company Overview
- Financial Insights
- Product/ Services Offerings
- Strategic Developments
- SWOT Analysis
- Anixa Biosciences
- Company Overview
- Financial Insights
- Product/ Services Offerings
- Strategic Developments
- SWOT Analysis
- Autolus Therapeutics
- Company Overview
- Financial Insights
- Product/ Services Offerings
- Strategic Developments
- SWOT Analysis
- Cartesian Therapeutics Inc
- Company Overview
- Financial Insights
- Product/ Services Offerings
- Strategic Developments
- SWOT Analysis
- Novartis AG
- Company Overview
- Financial Insights
- Product/ Services Offerings
- Strategic Developments
- SWOT Analysis
- CARsgen Therapeutics Holdings Limited
- Company Overview
- Financial Insights
- Product/ Services Offerings
- Strategic Developments
- SWOT Analysis

