Companion Diagnostics Market Overview and Key Insights:
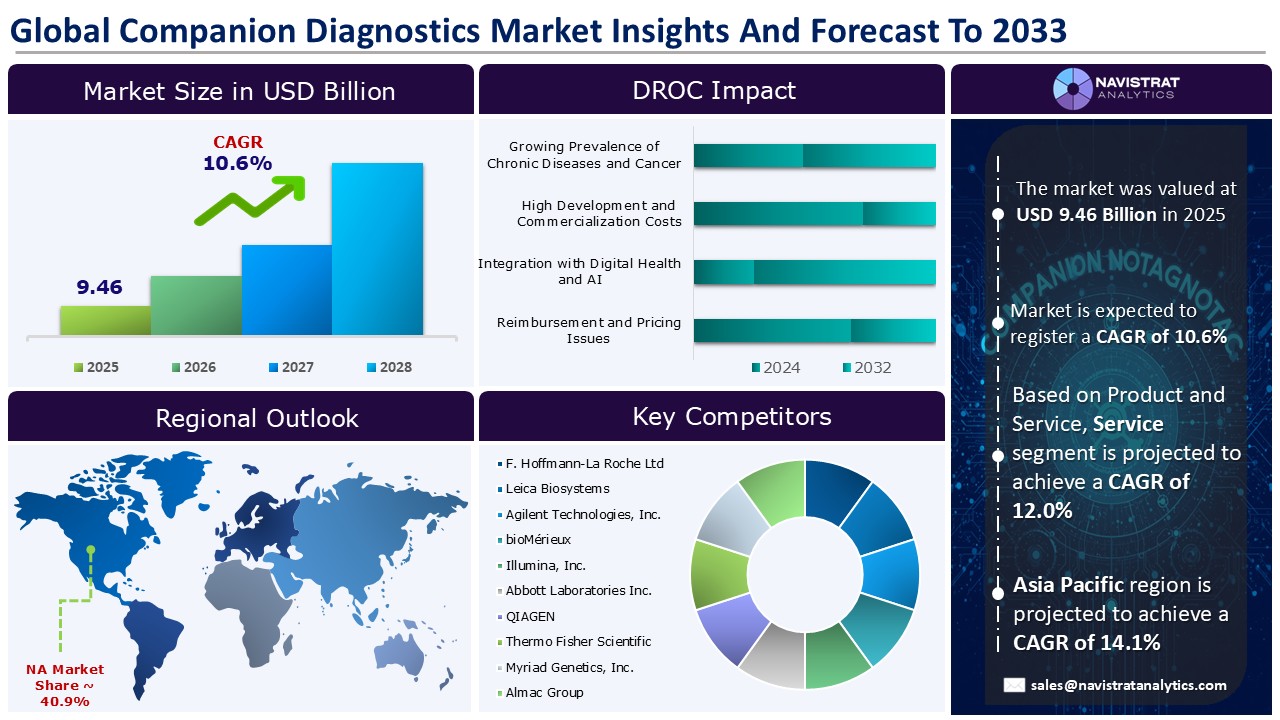
Companion diagnostics market size reached USD 9.46 Billion in 2025 and is expected to register a revenue CAGR of 10.6% during the forecast period. These are tests or assays that help identify patients who will benefit the most from a specific treatment or medicine. These tests are frequently used in precision medicine to adapt treatments to individual patients based on unique traits such as genetics, biomarker expression, or other diagnostic criteria.
Market Drivers:
Growing prevalence of chronic diseases and cancer is a key driver of revenue growth in companion diagnostics market. According to the American Cancer Society, estimates for lung cancer in the United States by 2025 are: approximately 226,650 new cases of lung cancer (110,680 males and 115,970 women). Approximately 124,730 fatalities from lung cancer (64,190 males and 60,540 women). Older persons are more likely to develop lung cancer. Most patients diagnosed with lung cancer are 65 or older; only a small minority are under the age of 45. The average age of those diagnosed is around 70. Lung cancer is the leading cause of cancer death in the United States, accounting for approximately one-fifth of all cancer deaths.
Companion diagnostic tests are created together with the therapeutic solution. This has the advantage of allowing for simultaneous approval based on the clinical benefit established in a pivotal registration trial for a particular therapeutic medication in a specific illness indication. With the development of next-generation DNA sequencing (NGS), it became possible to search for numerous markers in a single test at a reasonable cost. The approval of an NGS panel, which now includes companion diagnostic markers for 21 distinct oncology medications, enabled considerably more efficient testing for genetic markers in non-small cell lung cancer, melanoma, breast cancer, colorectal cancer, and ovarian cancer.
On October 2024, Roche has announced that the VENTANA CLDN18 RxDx Assay has been approved by the United States Food and Drug Administration (FDA) the first immunohistochemistry (IHC) companion diagnostic to detect CLDN18 protein expression in gastric or gastroesophageal junction (GEJ) adenocarcinoma tumors. This approval allows clinicians to identify patients who may be eligible for treatment with Astellas’ targeted therapy, VYLOY (zolbetuximab). The assay helps healthcare providers select individuals with gastric or GEJ adenocarcinoma who are suitable candidates for VYLOY, in line with the approved therapeutic product labeling.
Market Opportunity:
Integration with digital health and Artificial Intelligence (AI) acts as opportunities for companion diagnostics market. Digital pathology (DP) is increasingly being used in cancer diagnostics, providing new techniques for faster, higher-quality, and more accurate detection. The practice of diagnostic pathology has undergone a stunning transformation, with new tools such as digital imaging, advanced artificial intelligence (AI) algorithms, and computer-aided diagnostic techniques being used to assist, augment, and empower computational histopathology and AI-enabled diagnosis. However, in France, it is obligatory to test patients for dihydropyrimidine dehydrogenase deficiency prior to starting fluoropyrimidine treatment because the condition can place patients at high risk of serious effects, including death.
On January 2025, Tempus AI, Inc., a technology company that is pioneering the adoption of AI to promote precision medicine and patient care, has announced the national launch of its FDA-approved, NGS-based in vitro diagnostic equipment, xTCDx. xT CDx is now available to all ordering clinicians countrywide. xT CDx is an FDA-approved test that provides comprehensive insights using one of the most extensive reported gene panels available. xT CDx is a next-generation sequencing test with 648 genes for solid tumor profiling that includes microsatellite instability status and companion diagnostic claims for colorectal cancer patients.
Recent Trends:
Emerging trends include imaging-based companion diagnostics, Antibody-drug conjugates (ADCs), digital and AI-enabled diagnostics, integration with potential biomarkers, and expanding use in personalized medicine.
Imaging-guided companion diagnostics, such as MRI, PET, SPECT, and photoacoustic imaging, provide real-time, non-invasive, and dynamic insights into biomarker status in vivo, outperforming in vitro and ex vivo research techniques. Combining imaging-based companion diagnostics with radiation offers the potential to enable individualized dosage prescriptions based on real-time biomarker activity, addressing the issues posed by tumor heterogeneity and individual variability.
Furthermore, imaging-based CDx is critical for monitoring therapy outcomes. Healthcare professionals can evaluate the effectiveness of their chosen therapy and make required adjustments to improve patient outcomes by analyzing changes in disease features on a regular basis using imaging.
On July 2024, BD, a leading global medical technology company, and Quest Diagnostics, a diagnostic information services leader, have announced a global collaboration agreement to develop, manufacture, and commercialize flow cytometry-based companion diagnostics (CDx) to aid in the selection of the best treatment for cancer patients and others. BD and Quest will collaborate to offer the pharmaceutical industry an end-to-end solution for CDx development. The collaborative offers will span from exploratory panel creation to the production and sale of FDA-approved diagnostic kits.
Restraints & Challenges:
Companion diagnostic regulations aim to protect patients by assuring the quality and consistency of treatment-guided biomarker testing in clinical trials and clinical practice. However, present rules have had unforeseen negative repercussions for precision medicine innovation, implementation, and accessibility, raising complexity and expense burden, and impeding the development of innovative diagnostics and biomarker-targeted therapies.
Traditional companion diagnostics face a major limitation because they evaluate a narrow range of biomarkers and often require costly tissue samples when clinicians must run multiple single-biomarker tests to select the appropriate therapy. Recent research has also demonstrated that identifying a therapeutic target alone does not adequately predict drug efficacy; clinicians must supplement target detection with additional data to assess treatment resistance.
Product and Service Segment Insights and Analysis:
Based on the product and service segment, companion diagnostics market is segmented into product and service.
Product segment contributed the largest market share in 2025. It is further sub segmented into instrument, consumables, and software. CDx devices support clinical trials by enabling patient stratification, diagnosis, treatment selection, prognosis assessment, therapy response monitoring, and resistance marker evaluation. These devices play a critical role in therapeutic decision-making and in the development of precision medicine strategies. In clinical studies, the use of CDx devices often requires separate regulatory approval from relevant authorities such as an Institutional Review Board (IRB) or the U.S. Food and Drug Administration (FDA). The effectiveness of companion diagnostic tests depends on their reliability, the level of patient safety they ensure, and the speed at which they deliver results. As a result, developers subject CDx devices to regulatory and development processes that are as rigorous as those for the corresponding therapeutic agents, which are typically developed in parallel.
On August 2024, Illumina, Inc., a global leader in DNA sequencing and array-based technologies, announced that its TruSight Oncology (TSO) Comprehensive in vitro diagnostic (IVD) test and its first two companion diagnostic (CDx) indications have been approved by the FDA. This single test examines over 500 genes to profile a patient’s solid tumor, increasing the possibility of detecting an immuno-oncology biomarker or clinically actionable biomarkers that allow for targeted therapy or clinical trial enrollment.

Technology Segment Insights and Analysis:
Based on the technology, companion diagnostics market is segmented into Polymerase Chain Reaction (PCR), Next-Generation Sequencing (NGS), Fluorescence In Situ Hybridization (FISH), Immunohistochemistry (IHC), gene expression arrays, and others.
Next-Generation Sequencing (NGS) segment contributed the largest market share in 2025. It is further sub segmented into whole genome sequencing, targeted gene panels, and exome sequencing. NGS is a transformative tool in research and drug discovery, as well as clinical testing and diagnostics. The convergence of simultaneous technological development and regulatory and legislative reforms has resulted in a rapid acceptance of NGS in the clinic, with several NGS-based diagnostics receiving regulatory approval, including as companion and complementary diagnostics.
NGS provides great benefits in terms of low cost per sequenced base and high throughput, but it also has significant advantages in companion diagnostics and customized therapy. For example, most of the FDA-approved in-vitro diagnostic tests (IVD) detect only one or a few indicators. In comparison, when exploited to its full potential, NGS can offer more than three billion bases in the human genome and detect millions of genetic variants in a single test. This analytical power is particularly useful in companion diagnostics used in cancer therapy, where a rising number of mutations must be examined.
On June 2025, QIAGEN N.V. and Incyte have entered into a new global collaboration to develop a novel diagnostic panel that will support Incyte’s broad portfolio of investigational therapies for patients with myeloproliferative neoplasms (MPNs), a group of rare blood cancers. The collaboration specifically supports the development of Incyte’s monoclonal antibody INCA033989, which targets mutant calreticulin (mutCALR) and is under development for the treatment of myelofibrosis (MF) and essential thrombocythemia (ET). According to the Master Collaboration Agreement with Incyte, QIAGEN will create a multimodal panel employing next-generation sequencing (NGS) technology to discover clinically relevant gene changes in hematological malignancies.
Application Segment Insights and Analysis:
Based on the application, Companion Diagnostics market is segmented into cardiovascular diseases, oncology, infectious diseases, neurological diseases, autoimmune diseases, and others.
Oncology segment contributed the largest market share in 2025. It is further sub segmented into breast cancer, lung cancer, colorectal cancer, melanoma, prostate cancer, and others. According to National Brain Tumor Society, In 2023, a projected 94,390 Americans will be diagnosed with a new primary brain tumor. In 2023, it is anticipated that 67,440 will be non-malignant (benign). Non-malignant meningiomas are the most often occurring primary non-malignant brain tumors, accounting for 39.7% of all cancers and 55.4% of all non-malignant tumors. Glioblastoma is the most prevalent primary malignant brain tumor, accounting for 14.2% of all tumors and 50.1% of all malignant tumors.
On January 2025, Foundation Medicine, Inc. announced that the U.S. Food and Drug Administration (FDA) has approved FoundationOne CDx as a companion diagnostic for Day One Biopharmaceuticals’ OJEMDA, a type II RAF inhibitor, for the treatment of patients six months and older with relapsed or refractory pediatric low-grade glioma (pLGG) harboring a BRAF fusion or rearrangement, or BRAF V600 mutation. FoundationOne CDx is OJEMDA’s first and only companion diagnostic.
Geographical Outlook:
Companion diagnostics market is strategically segmented by geography to provide a comprehensive understanding of regional market dynamic. Discover demand analysis, emerging trends, and growth opportunities shaping market performance across different region and countries.
North America Companion Diagnostics Market:
North America holds the largest share of the companion diagnostics market in 2025. The region drives this dominance through the rising prevalence of chronic diseases and cancer, along with rapid advancements in molecular diagnostic technologies. According to the American Cancer Society, melanoma in the United States is expected to cause approximately 8,430 deaths by 2025, with around 104,960 new cases diagnosed. Having lighter skin is a significant risk factor for melanoma. Overall, the lifetime chance of developing melanoma is approximately 3% (1 in 33) for White individuals, 0.1% (1 in 1,000) for Black people, and 0.5% (1 in 200) for Hispanic people.
On June 2025, Randox Laboratories stated that the United States Food and Drug Administration (FDA) has approved the De Novo application for the company’s first Companion Diagnostic (CDx). The Randox ConcizuTrace ELISA (enzyme-linked immunosorbent assay) quantitatively measures concizumab-mtci concentrations in human 3.2% citrated plasma samples collected from hemophilia A and B patients four weeks after treatment initiation. Clinicians use the measured concizumab-mtci levels to guide dose adjustments in accordance with the drug label. The approved ConcizuTrace ELISA is part of the Global Randox ConcizuTrace ELISA Service Solutions.
Asia Pacific Companion Diagnostics Market:
Asia Pacific is expected to register the fastest growth rate during the forecasted period, driven by growing prevalence of chronic diseases and cancer and rising demand for personalized medicine. APAC accounts for roughly two-thirds of the world’s population. Rising disposable incomes are also driving up healthcare spending, thanks to a growing middle class across the area. Government measures are also fuelling the surge. The Healthy China 2030 plan intends to increase the size of the country’s health-care business to roughly USD 2.4 trillion by 2030, essentially doubling it in a decade. Moreover, Japan’s companion diagnostics market is rapidly expanding, driven by the country’s commitment to personalized medicine and rising demand for advanced diagnostic tools due to short approval timelines, a high likelihood of reimbursement approval, and clinical demand for companion diagnostics.
On July 2024, Riken Genesis Co., Ltd., Amoy Diagnostics Co., Ltd., and Precision Medicine Asia Co., Ltd. announced that the Japanese Ministry of Health, Labour and Welfare (MHLW) has approved the AmoyDx Pan Lung Cancer PCR Panel as a companion diagnostic for Haiyitan (gumarontinib), developed by Haihe Biopharma K.K. The 50 mg tablet form of Haiyitan received MHLW approval in June 2024 for the treatment of patients with unresectable advanced or recurrent non-small cell lung cancer (NSCLC) harboring MET exon 14 (METex14) skipping mutations.
Europe Companion Diagnostics Market:
Europe is expected to have considerable market share in 2025. Personalized medicine, a novel approach that tailors medical treatments to individual traits, is gaining traction in Europe. As genomes and molecular diagnostics progress, pharmaceutical corporations are poised to lead a dramatic shift in healthcare. On August 2024, QIAGEN has announced the expansion of its Master Collaboration Agreement with AstraZeneca to develop and commercialize companion diagnostics (CDx) for AstraZeneca’s future treatments addressing chronic diseases. Under the terms of the agreement, QIAGEN will create and validate a genotyping assay using its syndromic testing platform, QIAstat-Dx. The test will allow specialist care providers to possibly undertake genotyping while patients are receiving normal clinical examinations, allowing for faster decision-making on the applicability of AstraZeneca’s genomically targeted medicines.

Competition Analysis:
Companion diagnostics market is characterized by a fragmented structure, with several players competing across various segments and regions. list of major players included in companion diagnostics market report are:
- Hoffmann-La Roche Ltd
- Leica Biosystems
- Agilent Technologies, Inc.
- bioMérieux
- Illumina, Inc.
- Abbott Laboratories Inc.
- QIAGEN
- Thermo Fisher Scientific
- Myriad Genetics, Inc.
- Almac Group
- Tempus AI, Inc.
- Foundation Medicine, Inc.
- BD
Strategic Developments in Companion Diagnostics Market:
- In June 2025, GeneCentric Therapeutics, a startup that uses gene expression to improve precision medicine, announced the initial close of a USD 8.0 million Series C funding. The funds will be used to launch and commercialize GeneCentric’s GenomicsNext, the first integrated platform for comprehensive liquid biopsy testing that provides thousands of gene expression measurements and high-fidelity DNA variant detection from circulating tumor DNA (ctDNA) in a single sample.
- In June 2025, Foresight Diagnostics, a prominent diagnostics company focused on ultra-sensitive minimal residual disease (MRD) detection, announced a strategic global partnership with QIAGEN to develop and commercialize a kit-based version of its CLARITY assay. The aim of this collaboration is to expand the use of the assay for in vitro diagnostic (IVD) and companion diagnostic (CDx) applications worldwide, particularly in lymphoma and other hematological cancers. This partnership leverages Foresight’s cutting-edge MRD detection technology alongside QIAGEN’s extensive expertise in molecular diagnostic development and global commercialization.
- In April 2024, Labcorp, a global leader in innovative and comprehensive laboratory services, announced that the United States Food and Drug Administration (FDA) has approved its nAbCyte Anti-AAVRh74var HB-FE Assay, a companion diagnostic (CDx) used to determine patient eligibility for treatment with BEQVEZ (fidanacogene elaparvovec-dzkt), Pfizer’s recently FDA-approved hemophilia B gene therapy. The nAbCyte cell-based neutralizing antibody assay is part of Pfizer’s program to tailor recombinant adeno-associated virus (rAAV)-based gene treatments to specific patient populations.
Key Advantages for Stakeholders:
Navistrat Analytics’ industry report provides an in-depth quantitative analysis of various market segments, historical and current trends, market forecasts, and dynamics within the global market. The historical years covered in this report are 2023 to 2024, with 2025 serving as the base year for market size calculations. The forecast period extends from 2026 to 2033.
The report includes an executive summary and a comprehensive overview of market drivers, restraints, opportunities, and challenges (DROC), along with insights into regulatory standards. It features detailed analyses such as PORTER’s Five Forces, SWOT, and PESTLE, as well as assessments of technological trends and the competitive landscape.
PORTER’s Five Forces analysis helps stakeholders evaluate the impact of new entrants, competitive rivalry, supplier power, buyer power, and substitution threats, enabling them to assess the level of competition and the attractiveness of the global market. The competitive landscape provides stakeholders with a clear understanding of the current market positions of key players, offering valuable insights into their competitive environment.
Scope And Key Highlights Of Companion Diagnostics Market Report:
| Report Features | Details |
| Market Size in 2025 | USD 9.46 Billion |
| Market Growth Rate in CAGR (2026-2033) | 10.6% |
| Market Revenue forecast to 2033 | USD 21.28 Billion |
| Base year | 2025 |
| Historical year | 2023-2024 |
| Forecast period | 2026-2033 |
| Report Pages | 450 |
| Segments covered |
|
| Regional scope |
|
| Country Scope |
|
| Key Market Players |
|
| Delivery Format | Reports are delivered in PDF format via email. |
| Customization scope | Request for Customization |
Companion Diagnostics market report offers a detailed analysis of market size, including historical revenue (in USD Billion) data for 2023-2024 and revenue forecasts for 2026-2033 across the following segments:
- Product and Service Outlook (Revenue, USD Billion; 2023-2033)
- Product
- Instrument
- Consumables
- Software
- Service
- Product
- Technology Outlook (Revenue, USD Billion; 2023-2033)
- Polymerase Chain Reaction (PCR)
- Real-time PCR
- Multiplex PCR
- Reverse Transcription PCR
- Next-Generation Sequencing (NGS)
- Whole Genome Sequencing
- Targeted Gene Panels
- Exome Sequencing
- Fluorescence In Situ Hybridization (FISH)
- Immunohistochemistry (IHC)
- Gene Expression Arrays
- Others
- Polymerase Chain Reaction (PCR)
- Indication Outlook (Revenue, USD Billion; 2023-2033)
- Cardiovascular Diseases
- Oncology
- Breast Cancer
- Lung Cancer
- Colorectal Cancer
- Melanoma
- Prostate Cancer
- Others
- Infectious Diseases
- HIV/AIDS
- Hepatitis
- Others
- Neurological Diseases
- Alzheimer’s Disease
- Parkinson’s Disease
- Others
- Autoimmune Diseases
- Rheumatoid Arthritis
- Systemic Lupus Erythematosus
- Others
- Others
- Application Outlook (Revenue, USD Billion; 2023-2033)
- Therapeutic Monitoring
- Drug Response Prediction
- Dose Optimization
- Risk Assessment
- Disease Diagnosis
- Drug Development
- Clinical Trials
- Drug Efficacy Studies
- Therapeutic Monitoring
- Application Outlook (Revenue, USD Billion; 2023-2033)
- Hospitals and Clinics
- Diagnostic Laboratories
- Academic and Research Institutions
- Biopharmaceutical Companies
- Regional Outlook (Revenue, USD Billion; 2023-2033)
- North America
- U.S.
- Canada
- Mexico
- Europe
- Germany
- France
- U.K.
- Italy
- Spain
- Benelux
- Nordic Countries
- Rest of Europe
- Asia Pacific
- China
- India
- Japan
- South Korea
- Oceania
- ASEAN Countries
- Rest of APAC
- Latin America
- Brazil
- Rest of LATAM
- Middle East & Africa
- GCC Countries
- South Africa
- Israel
- Turkey
- Rest of MEA
- North America
Frequently Asked Questions (FAQ) about Companion Diagnostics market report
The market size of companion diagnostics market was 9.46 billion in 2025.
The market size of companion diagnostics market is expected to register compound annual growth rate (CAGR) of 10.6% over the forecast period.
Growing prevalence of chronic diseases and cancer, rising demand for personalized medicine, and technological advancements in molecular diagnostic technologies are major key factors driving the market revenue growth of companion diagnostics market.
High development and commercialization costs and complex regulatory pathways are key limiting factors driving the market.
Asia Pacific account for fastest revenue growth of 14.1%.
Product is the major leading segment of companion diagnostics market in terms of product and service.
- Market Definition
- Research Objective
- Research Methodology
- Research Design
- Data Collection Methods
- Primary
- Secondary
- Market Size Estimation
- Top-down Indication
- Bottom-up Indication
- Forecasting Methodology
- Tools and Models Used
- Market Overview and Trends
- Market Size and Forecast
- Industry Analysis
- Market Driver, Restraints, Opportunity, and Challenges (DROC) Analysis
- Market Drivers
- Growing prevalence of chronic diseases and cancer
- Rising demand for personalized medicine
- Technological advancements in molecular diagnostic technologies
- Market Restraints
- High development and commercialization costs
- Complex regulatory pathways
- Market Opportunities
- Integration with digital health and AI
- Liquid biopsy and non-invasive testing
- Expansion of into infectious diseases, cardiology, neurology, and autoimmune disorders
- Market Challenges
- Reimbursement and pricing issues
- Rapid mutation of target diseases
- Regulatory Landscape
- North America
- Europe
- Asia Pacific
- Latin America
- Middle East and Africa
- Strategic Insights
- Porter’s Five Forces Analysis
- PESTLE Analysis
- Price Trend Analysis
- Value Chain Analysis
- Technological Trends
- Recent Developments
- Funding
- Merger and Acquisition
- Expansion
- Partnership and Collaboration
- Product/ Service Launch
- Product and Service Market Revenue Estimates and Forecasts, 2023-2033
- Product
- Instrument
- Consumables
- Software
- Service
- Product
- Technology Market Revenue Estimates and Forecasts, 2023-2033
- Polymerase Chain Reaction (PCR)
- Real-time PCR
- Multiplex PCR
- style=”font-family: arial, helvetica, sans-serif;”>Reverse Transcription PCR
- Toxicology Testing
- Whole Genome Sequencing
- Targeted Gene Panels
- Exome Sequencing
- Fluorescence In Situ Hybridization (FISH)
- Immunohistochemistry (IHC)
- Gene Expression Arrays
- Others
- Polymerase Chain Reaction (PCR)
- Indication Market Revenue Estimates and Forecasts, 2023-2033
- Cardiovascular Diseases
- Oncology
- Breast Cancer
- Lung Cancer
- Colorectal Cancer
- Melanoma
- Prostate Cancer
- Others
- Infectious Diseases
- HIV/AIDS
- Hepatitis
- Others
- Neurological Diseases
- Alzheimer’s Disease
- Parkinson’s Disease
- Others
- Autoimmune Diseases
- Rheumatoid Arthritis
- Systemic Lupus Erythematosus
- Others
- Others
- Application Market Revenue Estimates and Forecasts, 2023-2033
- Therapeutic Monitoring
- Drug Response Prediction
- Dose Optimization
- Risk Assessment
- Disease Diagnosis
- Drug Development
- Clinical Trials
- Drug Efficacy Studies
- Therapeutic Monitoring
- End-Use Market Revenue Estimates and Forecasts, 2023-2033
- Hospitals and Clinics
- Diagnostic Laboratories
- Academic and Research Institutions
- Biopharmaceutical Companies
- Companion Diagnostics Market Revenue Estimates and Forecasts by Region, 2023-2033, USD Billion
- North America
- North America Companion Diagnostics Market By Product and Service, Market Revenue Estimates and Forecasts, 2023-2033, USD Billion
- Product
- Instrument
- Consumables
- Software
- Service
- Product
- North America Companion Diagnostics Market By Technology, Market Revenue Estimates and Forecasts, 2023-2033, USD Billion
- Polymerase Chain Reaction (PCR)
- Real-time PCR
- Multiplex PCR
- Reverse Transcription PCR
- Next-Generation Sequencing (NGS)
- Whole Genome Sequencing
- Targeted Gene Panels
- Exome Sequencing
- Fluorescence In Situ Hybridization (FISH)
- Immunohistochemistry (IHC)
- Gene Expression Arrays
- Others
- Polymerase Chain Reaction (PCR)
- North America Companion Diagnostics Market By Indication, Market Revenue Estimates and Forecasts, 2023-2033, USD Billion
- Cardiovascular Diseases
- Oncology
- Breast Cancer
- Lung Cancer
- Colorectal Cancer
- Melanoma
- Prostate Cancer
- Others
- Infectious Diseases
- HIV/AIDS
- Hepatitis
- Others
- Neurological Diseases
- Alzheimer’s Disease
- Parkinson’s Disease
- Others
- Autoimmune Diseases
- Rheumatoid Arthritis
- Systemic Lupus Erythematosus
- Others
- Others
- North America Companion Diagnostics Market By Application, Market Revenue Estimates and Forecasts, 2023-2033, USD Billion
- Therapeutic Monitoring
- Drug Response Prediction
- Dose Optimization
- Risk Assessment
- Disease Diagnosis
- Drug Development
- Clinical Trials
- Drug Efficacy Studies
- Therapeutic Monitoring
- North America Companion Diagnostics Market By End-Use, Market Revenue Estimates and Forecasts, 2023-2033, USD Billion
- Hospitals and Clinics
- Diagnostic Laboratories
- Academic and Research Institutions
- Biopharmaceutical Companies
- North America Companion Diagnostics Market Revenue Estimates and Forecasts by Country, 2023-2033, USD Billion
- United States
- Canada
- Mexico
- North America Companion Diagnostics Market By Product and Service, Market Revenue Estimates and Forecasts, 2023-2033, USD Billion
- Europe
- Europe Companion Diagnostics Market By Product and Service, Market Revenue Estimates and Forecasts, 2023-2033, USD Billion
- Product
- Instrument
- Consumables
- Software
- Service
- Product
- Europe Companion Diagnostics Market By Technology, Market Revenue Estimates and Forecasts, 2023-2033, USD Billion
- Polymerase Chain Reaction (PCR)
- Real-time PCR
- Multiplex PCR
- Reverse Transcription PCR
- Next-Generation Sequencing (NGS)
- Whole Genome Sequencing
- Targeted Gene Panels
- Exome Sequencing
- Fluorescence In Situ Hybridization (FISH)
- Immunohistochemistry (IHC)
- Gene Expression Arrays
- Others
- Polymerase Chain Reaction (PCR)
- Europe Companion Diagnostics Market By Indication, Market Revenue Estimates and Forecasts, 2023-2033, USD Billion
- Cardiovascular Diseases
- Oncology
- Breast Cancer
- Lung Cancer
- Colorectal Cancer
- Melanoma
- Prostate Cancer
- Others
- Infectious Diseases
- HIV/AIDS
- Hepatitis
- Others
- Neurological Diseases
- Alzheimer’s Disease
- Parkinson’s Disease
- Others
- Autoimmune Diseases
- Rheumatoid Arthritis
- Systemic Lupus Erythematosus
- Others
- Others
- Europe Companion Diagnostics Market By Application, Market Revenue Estimates and Forecasts, 2023-2033, USD Billion
- Therapeutic Monitoring
- Drug Response Prediction
- Dose Optimization
- Risk Assessment
- Disease Diagnosis
- Drug Development
- Clinical Trials
- Drug Efficacy Studies
- Therapeutic Monitoring
- Europe Companion Diagnostics Market By End-Use, Market Revenue Estimates and Forecasts, 2023-2033, USD Billion
- Hospitals and Clinics
- Diagnostic Laboratories
- Academic and Research Institutions
- Biopharmaceutical Companies
- Europe Companion Diagnostics Market Revenue Estimates and Forecasts by Country, 2023-2033, USD Billion
- Germany
- United Kingdom
- France
- Italy
- Spain
- Benelux
- Nordic Countries
- Rest of Europe
- Europe Companion Diagnostics Market By Product and Service, Market Revenue Estimates and Forecasts, 2023-2033, USD Billion
- Asia Pacific
- Asia Pacific Companion Diagnostics Market By Product and Service, Market Revenue Estimates and Forecasts, 2023-2033, USD Billion
- Product
- Instrument
- Consumables
- Software
- Service
- Product
- Asia Pacific Companion Diagnostics Market By Technology, Market Revenue Estimates and Forecasts, 2023-2033, USD Billion
- Polymerase Chain Reaction (PCR)
- Real-time PCR
- Multiplex PCR
- Reverse Transcription PCR
- Next-Generation Sequencing (NGS)
- Whole Genome Sequencing
- Targeted Gene Panels
- Exome Sequencing
- Fluorescence In Situ Hybridization (FISH)
- Immunohistochemistry (IHC)
- Gene Expression Arrays
- Others
- Polymerase Chain Reaction (PCR)
- Asia Pacific Companion Diagnostics Market By Indication, Market Revenue Estimates and Forecasts, 2023-2033, USD Billion
- Cardiovascular Diseases
- Oncology
- Breast Cancer
- Lung Cancer
- Colorectal Cancer
- Melanoma
- Prostate Cancer
- Others
- Infectious Diseases
- HIV/AIDS
- Hepatitis
- Others
- Neurological Diseases
- Alzheimer’s Disease
- Parkinson’s Disease
- Others
- Autoimmune Diseases
- Rheumatoid Arthritis
- Systemic Lupus Erythematosus
- Others
- Others
- Asia Pacific Companion Diagnostics Market By Application, Market Revenue Estimates and Forecasts, 2023-2033, USD Billion
- Therapeutic Monitoring
- Drug Response Prediction
- Dose Optimization
- Risk Assessment
- Disease Diagnosis
- Drug Development
- Clinical Trials
- Drug Efficacy Studies
- Therapeutic Monitoring
- Asia Pacific Companion Diagnostics Market By End-Use, Market Revenue Estimates and Forecasts, 2023-2033, USD Billion
- Hospitals and Clinics
- Diagnostic Laboratories
- Academic and Research Institutions
- Biopharmaceutical Companies
- Asia Pacific Companion Diagnostics Market Revenue Estimates and Forecasts by Country, 2023-2033, USD Billion
- China
- India
- Japan
- South Korea
- Oceania
- ASEAN Countries
- Rest of Asia-Pacific
- Asia Pacific Companion Diagnostics Market By Product and Service, Market Revenue Estimates and Forecasts, 2023-2033, USD Billion
- Latin America
- Latin America Companion Diagnostics Market By Product and Service, Market Revenue Estimates and Forecasts, 2023-2033, USD Billion
- Product
- Instrument
- Consumables
- Software
- Service
- Product
- Latin America Companion Diagnostics Market By Technology, Market Revenue Estimates and Forecasts, 2023-2033, USD Billion
- Polymerase Chain Reaction (PCR)
- Real-time PCR
- Multiplex PCR
- Reverse Transcription PCR
- Next-Generation Sequencing (NGS)
- Whole Genome Sequencing
- Targeted Gene Panels
- Exome Sequencing
- Fluorescence In Situ Hybridization (FISH)
- Immunohistochemistry (IHC)
- Gene Expression Arrays
- Others
- Polymerase Chain Reaction (PCR)
- Latin America Companion Diagnostics Market By Indication, Market Revenue Estimates and Forecasts, 2023-2033, USD Billion
- Cardiovascular Diseases
- Oncology
- Breast Cancer
- Lung Cancer
- Colorectal Cancer
- Melanoma
- Prostate Cancer
- Others
- Infectious Diseases
- HIV/AIDS
- Hepatitis
- Others
- Neurological Diseases
- Alzheimer’s Disease
- Parkinson’s Disease
- Others
- Autoimmune Diseases
- Rheumatoid Arthritis
- Systemic Lupus Erythematosus
- Others
- Others
- Latin America Companion Diagnostics Market By Application, Market Revenue Estimates and Forecasts, 2023-2033, USD Billion
- Therapeutic Monitoring
- Drug Response Prediction
- Dose Optimization
- Risk Assessment
- Disease Diagnosis
- Drug Development
- Clinical Trials
- Drug Efficacy Studies
- Therapeutic Monitoring
- Latin America Companion Diagnostics Market By End-Use, Market Revenue Estimates and Forecasts, 2023-2033, USD Billion
- Hospitals and Clinics
- Diagnostic Laboratories
- Academic and Research Institutions
- Biopharmaceutical Companies
- Latin America Companion Diagnostics Market Revenue Estimates and Forecasts by Country, 2023-2033, USD Billion
- Brazil
- Rest of Latin America
- Latin America Companion Diagnostics Market By Product and Service, Market Revenue Estimates and Forecasts, 2023-2033, USD Billion
- Middle East and Africa
- Middle East and Africa Companion Diagnostics Market By Product and Service, Market Revenue Estimates and Forecasts, 2023-2033, USD Billion
- Product
- Instrument
- Consumables
- Software
- Service
- Product
- Middle East and Africa Companion Diagnostics Market By Technology, Market Revenue Estimates and Forecasts, 2023-2033, USD Billion
- Polymerase Chain Reaction (PCR)
- Real-time PCR
- Multiplex PCR
- Reverse Transcription PCR
- Next-Generation Sequencing (NGS)
- Whole Genome Sequencing
- Targeted Gene Panels
- Exome Sequencing
- Fluorescence In Situ Hybridization (FISH)
- Immunohistochemistry (IHC)
- Gene Expression Arrays
- Others
- Polymerase Chain Reaction (PCR)
- Middle East and Africa Companion Diagnostics Market By Indication, Market Revenue Estimates and Forecasts, 2023-2033, USD Billion
- Cardiovascular Diseases
- Oncology
- Breast Cancer
- Lung Cancer
- Colorectal Cancer
- Melanoma
- Prostate Cancer
- Others
- Infectious Diseases
- HIV/AIDS
- Hepatitis
- Others
- Neurological Diseases
- Alzheimer’s Disease
- Parkinson’s Disease
- Others
- Autoimmune Diseases
- Rheumatoid Arthritis
- Systemic Lupus Erythematosus
- Others
- Others
- Middle East and Africa Companion Diagnostics Market By Application, Market Revenue Estimates and Forecasts, 2023-2033, USD Billion
- Therapeutic Monitoring
- Drug Response Prediction
- Dose Optimization
- Risk Assessment
- Disease Diagnosis
- Drug Development
- Clinical Trials
- Drug Efficacy Studies
- Therapeutic Monitoring
- Middle East and Africa Companion Diagnostics Market By End-Use, Market Revenue Estimates and Forecasts, 2023-2033, USD Billion
- Hospitals and Clinics
- Diagnostic Laboratories
- Academic and Research Institutions
- Biopharmaceutical Companies
- Middle East and Africa Companion Diagnostics Market Revenue Estimates and Forecasts by Country, 2023-2033, USD Billion
- GCC Countries
- South Africa
- Israel
- Turkey
- Rest of Middle East & Africa
- Middle East and Africa Companion Diagnostics Market By Product and Service, Market Revenue Estimates and Forecasts, 2023-2033, USD Billion
- North America
- Market Share Analysis
- Revenue Market Share by Key Players (2023-2024)
- Analysis of Top Players by Market Presence
- <span style=”font-family: arial, helvetica, sans-serif;”>Competitive Matrix
- Competitive Strategies
- Mergers and Acquisitions
- Partnerships and Collaboration
- Investment and Fundings
- Agreement
- Expansion
- New Product/ Services Launches
- Technological Innovations
- Hoffmann-La Roche Ltd
-
- Company Overview
-
- Financial Insights
- Product and Service/ Services Offerings
- Strategic Develop
-
- ments
- SWOT Analysis
- Leica Biosystems
- Company Overview
- Financial Insights
- Product and Service/ Services Offerings
- Strategic Developments
- SWOT Analysis
- Agilent Technologies, Inc.
- Company Overview
- Financial Insights
- Product and Service/ Services Offerings
- Strategic Developments
- SWOT Analysis
- bioMérieux
- Company Overview
- Financial Insights
- Product and Service/ Services Offerings
- Strategic Developments
- SWOT Analysis
- Illumina, Inc.
- Company Overview
- Financial Insights
- Product and Service/ Services Offerings
- Strategic Developments
- SWOT Analysis
- Abbott Laboratories Inc.
- Company Overview
- Financial Insights
- Product and Service/ Services Offerings
- Strategic Developments
- SWOT Analysis
- QIAGEN
- Company Overview
- Financial Insights
- Product and Service/ Services Offerings
- Strategic Developments
- SWOT Analysis
- Thermo Fisher Scientific
- Company Overview
- Financial Insights
- Product and Service/ Services Offerings
- Strategic Developments
- SWOT Analysis
- Myriad Genetics, Inc.
- Company Overview
- Financial Insights
- Product and Service/ Services Offerings
- Strategic Developments
- SWOT Analysis
- Almac Group
- Company Overview
- Financial Insights
- Product and Service/ Services Offerings
- Strategic Developments
- SWOT Analysis
- Tempus AI, Inc.
- Company Overview
- Financial Insights
- Product and Service/ Services Offerings
- Strategic Developments
- SWOT Analysis
- Foundation Medicine, Inc.
- Company Overview
- Financial Insights
- Product and Service/ Services Offerings
- Strategic Developments
- SWOT Analysis
- BD
- Company Overview
- Financial Insights
- Product and Service/ Services Offerings
- Strategic Developments
- SWOT Analysis

