Preclinical contract research organization (CRO) Market Overview and Key Insights:
Preclinical Contract Research Organization (CRO) market size reached USD 8.79 Billion in 2024 and is expected to register a revenue CAGR of 12.7% during the forecast period. A preclinical CRO is a specialist organization that helps pharmaceutical and biotechnology businesses with early-stage drug development. Preclinical CROs provide a variety of services to bridge the gap between drug discovery and clinical trials.

Market Drivers:
Growing reliance on outsourcing by small and virtual biotech is a key driver of revenue growth in preclinical Contract Research Organization (CRO) market. Pharmaceutical and biotechnology businesses can outsource their research and development efforts to CROs to varying degrees, depending on their needs. Outsourcing research activities allows pharmaceutical and biotechnology companies to focus on their core capabilities while saving time and money on infrastructure and human investments.
In the competitive pharmaceutical sector, operational efficiency accelerates the release of innovative medications and cures, providing a competitive edge and faster revenue generation. Partnering with CROs allows pharmaceutical companies to adapt to changing market needs and manage their R&D activities more easily than maintaining in-house research and development centers, which require significant investments in infrastructure, personnel, and fixed costs.
Advancements in pharmaceutical technology, including high-throughput screening, molecular profiling, next-generation sequencing, bioinformatics analysis, and enhanced imaging, are driving the preclinical CRO market. Advances in genomics, proteomics, and other molecular profiling techniques have enabled personalized medicine approaches for medication development.
On June 2025, Samsung Biologics, a leading contract development and manufacturing organization (CDMO), has announced the launch of Samsung Organoids, which provides enhanced drug screening services to help clients with drug discovery and development. Samsung Organoids use data-driven analysis of candidate molecules to assist precision screening, which predicts patient responses, streamlines preclinical development, and accelerates timeframes for investigational new drug (IND). Samsung Biologics now offers preclinical research services, including target discovery, lead selection, preclinical development, and clinical trial preparation.
Market Opportunity:
Implementing AI-driven predictive toxicology and digital platforms acts as opportunities for preclinical contract research organization (CRO) market. AI-based solutions are increasingly being used in a variety of preclinical research and clinical trial settings. These approaches help CROs, pharmaceutical, biotechnology, and medical device businesses accelerate drug development processes, improve clinical trial efficacy, automate tests, and improve data analysis, management, and collecting accuracy. The ability to predict using AI-based models enables CROs to process and identify patterns in huge volumes of data, tailoring real-time updates and responding beyond human capabilities. Furthermore, the use of AI-powered chatbots can answer potential patients’ inquiries about clinical trials, increase credibility, and help them enroll.
On May 2025, Inductive Bio, a technology business that democratizes artificial intelligence (AI) models to change small molecule drug development, announced that it has raised USD 25 million in Series A funding. Inductive plans to use the funds to extend its AI model R&D, grow its pre-competitive data consortium, and spread its technology across the sector, leveling the playing field for both new startups and established pharmaceutical corporations.
Recent Trends:
Emerging trends include Artificial Intelligence (AI) & digital technologies in preclinical research, advanced bioanalytical platforms, adoption of organ-on-chip and 3D cell culture models, automation & high-throughput screening, and advanced imaging & in-vivo technologies.
The convergence of microfabrication and tissue engineering led in the creation of organ-on-a-chip technology, which provides an alternative to standard preclinical drug testing models. Organ-on-a-chip technologies’ capacity to imitate critical human physiological activities required for understanding medication effects improves preclinical safety and efficacy testing. Organ-on-a-chip technology has the potential to significantly enhance preclinical testing success rates, allowing for better prediction of how the medicine will perform in clinical trials.
On April 2023, Dynamic42 GmbH has begun the production of a new generation of unique biotech chips for organ-on-chip technology. The effect of chemical substances on human organ replicas can be investigated directly using this chip, which was designed internally. Furthermore, complicated immunological models can be mapped for application in fundamental research and medication development. Until far, designing the technical integration of spheroids (spherical tissue structures) on chip shapes with a simultaneously flowing culture media has proven difficult.
Restraints & Challenges:
Despite the benefits of outsourcing, the CRO business faces obstacles such as establishing dependable relationships and adapting to quick industry changes. CROs and drug research businesses may find it difficult to navigate the complex and frequently changing regulatory landscapes across different geographies. As laws become stricter, the compliance burden increases, resulting in longer deadlines and higher costs.
The costs of clinical trials and drug development continue to climb, driven by the demand for modern technology, comprehensive quality assurance, and adherence to global standards. Financial constraints might limit the breadth and extent of research projects. Furthermore, the dearth of competent specialists in the CRO industry is a considerable impediment. CROs face rising competition from biotech and pharmaceutical corporations for talented scientists and specialists, making attracting and maintaining staff more difficult.
Product Segment Insights and Analysis:
Based on the product, preclinical Contract Research Organization (CRO) market is segmented into instruments, accessories and consumables, and software.
Instruments segment contributed the largest market share in 2024. It is further sub segmented into microplate readers, flow cytometers, Micro-PET/CT, Immunohistochemistry (IHC) staining systems, High-performance liquid chromatography (HPLC) systems, mass spectrometers, and others. Preclinical CROs conduct all the product development required to prepare a therapy for clinical trials, making them critical to determining the safety and efficacy of a potential drug before it enters human clinical trials. Preclinical CROs do thorough testing and safety assessments employing pharmacology, toxicology, pharmacokinetics, and pharmacodynamics. These studies assess the possible hazards and dangers that the treatment may pose when provided on a large scale, including carcinogenicity, mutagenicity, reproductive toxicity, allergic or inflammatory reactions, bioavailability, and biocompatibility.
With the growing demand for high-throughput studies, automated systems have emerged as a critical component in modern labs. Automation speeds up repetitious operations, minimizes human error, improves accuracy, and simplifies workflow. Automated technology ensures productivity and uniformity in CROs, which frequently deal with massive data sets and rapid turnaround times.
On July 2025, Auriga Research Private Limited, one of India’s leading Contract Research Organizations (CROs), has inked a strategic Memorandum of Understanding (MoU) with the San Francisco Research Institute (SFRI), a major US-based organization in healthcare research and innovation. This historic alliance intends to leverage both organisations’ core skills to boost AI-integrated clinical research, expedite regulatory routes, and accelerate the development and market access of wellness and healthcare products in India, North America, and Africa.
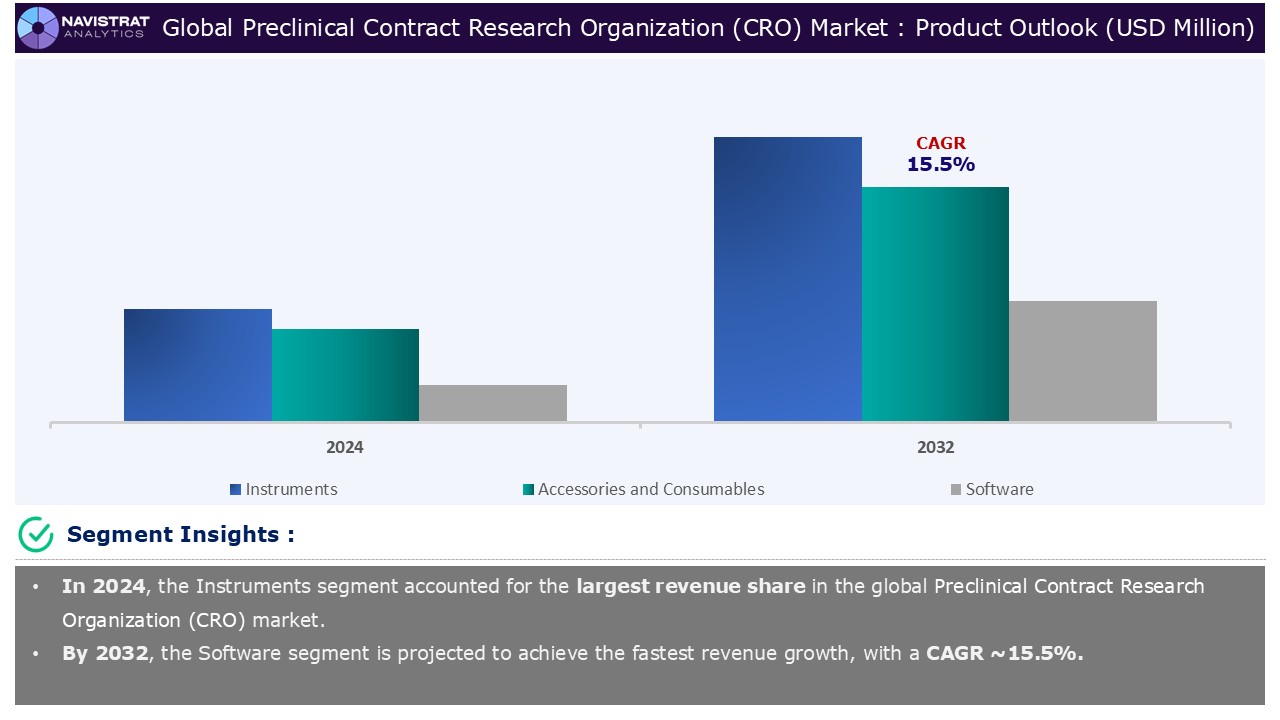
Service Type Segment Insights and Analysis:
Based on the service type, Preclinical Contract Research Organization (CRO) market is segmented into bioanalysis and DMPK studies, toxicology testing, compound management, chemistry, safety pharmacology, and others.
Bioanalysis and DMPK studies segment contributed the largest market share in 2024. It is further sub segmented into in vitro ADME and in-vivo PK. DMPK studies enable drug developers to experimentally examine a drug candidate’s intrinsic qualities to ensure that it can and will be removed from the body when delivered to a patient without forming hazardous byproducts, reaching dangerous exposure levels, or causing undesirable side effects. These new entities operate fundamentally differently from classic small molecules, which may be defined using a very standard set of assays for absorption, distribution, metabolism, and excretion (ADME). Radiolabeled drugs, which are routinely employed in in vivo investigations but not in vitro, allow drug developers to correctly identify metabolites and determine how much of each is present in plasma or excreta.
On April 2025, QPS, a leading global contract research organization (CRO) that specializes in preclinical and clinical drug development services, has expanded its pharmacology disease model portfolio to include highly relevant and sophisticated models for metabolic dysfunction-associated steatohepatitis (MASH), ulcerative colitis (UC), wound healing, and psoriasis. These novel illness models give the biomedical community additional tools for evaluating possible therapy candidates in preclinical trials, opening the path for new advances in treatment.
Application Segment Insights and Analysis:
Based on the Application, Preclinical Contract Research Organization (CRO) market is segmented into oncology, neurology, cardiology, infectious disease, metabolic disease, and others.
Oncology segment contributed the largest market share in 2024. According to American Cancer Society, excluding non-melanoma skin cancer, at least 40% of newly diagnosed malignancies in US adults (811,000 cases in 2025) are theoretically avoidable. Cigarette smoking, excess body weight, and alcohol intake account for 19%, 8%, and 5%, respectively. In 2025, the US is expected to have 316,950 new cases of invasive breast cancer in women and 2,800 in men, as well as 59,080 cases of ductal carcinoma in situ (DCIS) in women. In 2025, a projected 42,680 people would die from breast cancer, with 42,170 women and 510 males.
On April 2025, Debiopharm Research & Manufacturing S.A., a Swiss-based global biopharmaceutical company dedicated to the cure of cancer and infectious diseases, and Oncodesign Services, a leading CRO specializing in drug discovery and preclinical services, announced the signing of a license agreement for the use of the AbYlink technology in preclinical services. AbYlink is a regioselective bioconjugation method that is perfect for creating conjugates for therapeutic and non-invasive diagnostic purposes.
Geographical Outlook:
Preclinical contract research organization (CRO) market is strategically segmented by geography to provide a comprehensive understanding of regional market dynamic. Discover demand analysis, emerging trends, and growth opportunities shaping market performance across different region and countries.
North America Preclinical contract research organization (CRO) Market:
North America is registered to have highest market share in preclinical contract research organization (CRO) market in 2024. Small and virtual biotech companies are increasingly relying on outsourcing, and the demand for specialized services in novel modalities is rising. CROs tailor their services to meet the specific needs of pharmaceutical and biotechnology industries. They often customize services based on the complexity of preclinical and clinical trials, the field of therapy, and the available resources. Outsourcing to a CRO saves sponsors time and money, accelerates drug development, and creates a network of expertise to support the product throughout its lifecycle.
On April 2024, Smithers, a major provider of testing, information, consultancy, and compliance services, has introduced its Generic Pharmacokinetic (PK) Assay. The Smithers team of specialist scientists created and developed the generic PK Assay to meet the growing demand for more customizable PK testing solutions in preclinical bioanalysis. This method could be confirmed using specific IgG1, IgG2, and IgG4 molecules found in mouse and cynomolgus monkey blood samples.
Asia Pacific Preclinical contract research organization (CRO) Market:
Asia Pacific is expected to register the fastest growth rate during the forecasted period, driven by rising demand for specialized services in novel modalities and advancements in bioanalytical technologies. Asia-Pacific countries have renowned medical institutions and experienced investigators who are well-prepared to perform rigorous clinical research. These institutions frequently work with global counterparts, creating a collaborative environment that promotes innovation and scientific interchange. Many contract research organizations (CROs) are investing extensively in the Asia-Pacific area due to its cost-effective environment and potential for faster clinical trials, which is a big advantage for US-based sponsors looking to grow abroad. Furthermore, the APAC region’s blend of growing and developed economies creates a unique opportunity to serve global research demands while also adjusting to local peculiarities, ensuring that data obtained is comprehensive and representative of varied populations.
On September 2024, Tigermed, a leading global provider of clinical research solutions for the whole lifecycle of global biopharmaceutical and medical device products, has announced the completion of its purchase of CRO Micron, Inc. Following the acquisition, Micron will be integrated into Tigermed’s global network as a significant business operating under Tigermed Japan. Tigermed will expand its medical imaging capabilities in Japan and Asia-Pacific by leveraging Micron’s experienced service staff and strong regional understanding, as well as its global operational structure.
Europe Preclinical contract research organization (CRO) Market:
Europe is expected to have considerable market share in 2024. European region has enormous potential for doing high-quality clinical research due to established centralized healthcare infrastructures, well-educated investigators, and a general high level of interest in the clinical research sector in the region. Europe is an appealing clinical development region due to the commercial size of the end market and the availability of big patient populations. It has also been a leader in biosimilar regulation, and hence many of these studies take place in Europe.
On August 2023, RQM+, a global leader in MedTech services, has announced the acquisition of CRO Kottmann, a prominent contract research organization (CRO) in Germany that is known for its great customer service, scientific clinical research, and quality. This substantial step reinforces RQM+’s commitment to strategic growth and presence in Europe, as well as its dominance in emerging MedTech markets. The addition of CRO Kottmann expands RQM+’s existing geographical reach in global operations while also providing comprehensive end-to-end support to clients from pre-clinical phases to product commercialization.

Competition Analysis:
Preclinical Contract Research Organization (CRO) market is characterized by a fragmented structure, with several players competing across various segments and regions. list of major players included in preclinical Contract Research Organization (CRO) market report are:
- Charles River Laboratories
- Jubilant Biosys Limited
- Thermo Fisher Scientific Inc.
- Clinfinite Solutions
- Biospective Inc.
- Altasciences
- Eurofins Advinus
- Revvity, Inc.
- REPROCELL Inc.
- QPS Holdings
- Crown Bioscience
- WuXi AppTec
- Laboratory Corporation of America Holdings
- Intertek Group Plc
- Medpace, Inc.
Strategic Developments in Preclinical contract research organization (CRO) Market:
- In August 2025, CorestemChemon, a leading GLP-certified preclinical CRO based in South Korea, has formed a strategic cooperation with ATG Lifetech, a biotechnology business that specializes in transcriptome analytics and organoid-based modeling. This alliance seeks to provide next-generation, precision-driven non-clinical solutions to pharmaceutical companies and biotech ventures in the United States, Europe, and Asia. ATG Lifetech has proved its capabilities through international collaborations, such with ACROBiosystems (Switzerland), and has been chosen as a core developer in various South Korean government-backed biopharma ventures.
- In September 2024, ReSync Bio, a scientific software supplier that accelerates preclinical R&D, today announced the company’s launch and a funding round from Caffeinated Capital and Atria Ventures. The ReSync technology simplifies cooperation between drug discovery teams and their labs, whether they are in-house or outsourced CROs. The platform features carefully regulated permissioning, allowing businesses to manage data access and cooperation while protecting intellectual property.
Key Advantages for Stakeholders:
Navistrat Analytics’ industry report provides an in-depth quantitative analysis of various market segments, historical and current trends, market forecasts, and dynamics within the global market. The historical years covered in this report are 2022 to 2023, with 2024 serving as the base year for market size calculations. The forecast period extends from 2025 to 2032.
The report includes an executive summary and a comprehensive overview of market drivers, restraints, opportunities, and challenges (DROC), along with insights into regulatory standards. It features detailed analyses such as PORTER’s Five Forces, SWOT, and PESTLE, as well as assessments of technological trends and the competitive landscape.
PORTER’s Five Forces analysis helps stakeholders evaluate the impact of new entrants, competitive rivalry, supplier power, buyer power, and substitution threats, enabling them to assess the level of competition and the attractiveness of the global market. The competitive landscape provides stakeholders with a clear understanding of the current market positions of key players, offering valuable insights into their competitive environment.
Scope And Key Highlights Of Preclinical Contract Research Organization (CRO) Market Report:
| Report Features | Details |
| Market Size in 2024 | USD 8.79 Billion |
| Market Growth Rate in CAGR (2025–2032) | 12.7% |
| Market Revenue forecast to 2032 | USD 22.98 Billion |
| Base year | 2024 |
| Historical year | 2022-2023 |
| Forecast period | 2025-2032 |
| Report Pages | 450 |
| Segments covered |
|
| Regional scope |
|
| Country Scope |
|
| Key Market Players |
|
| Delivery Format | Reports are delivered in PDF format via email. |
| Customization scope | Request for Customization |
Preclinical Contract Research Organization (CRO) market report offers a detailed analysis of market size, including historical revenue (in USD Billion) data for 2022-2023 and revenue forecasts for 2025-2032 across the following segments:
- Product Outlook (Revenue, USD Billion; 2022-2032)
- Instruments
- Microplate readers
- Flow cytometers
- Micro-PET/CT
- Immunohistochemistry (IHC) staining systems
- High-performance liquid chromatography (HPLC) systems
- Mass spectrometers
- Others
- Accessories and Consumables
- Reagents and Chemicals
- Cell Lines
- Antibodies
- Others
- Software
- Instruments
- Service Type Outlook (Revenue, USD Billion; 2022-2032)
- Bioanalysis and DMPK studies
- In vitro ADME
- In-vivo PK
- Toxicology Testing
- GLP
- Non-GLP
- Compound Management
- Process R&D
- Custom Synthesis
- Others
- Chemistry
- Medicinal Chemistry
- Computation Chemistry
- Safety Pharmacology
- Others
- Bioanalysis and DMPK studies
- Model Type Outlook (Revenue, USD Billion; 2022-2032)
- Patient Derived Organoid (PDO) Model
- Patient derived xenograft Model
- Application Outlook (Revenue, USD Billion; 2022-2032)
- Oncology
- Neurology
- Cardiology
- Infectious Disease
- Metabolic Disease
- Others
- Regional Outlook (Revenue, USD Billion; 2022-2032)
- North America
- U.S.
- Canada
- Mexico
- Europe
- Germany
- France
- U.K.
- Italy
- Spain
- Benelux
- Nordic Countries
- Rest of Europe
- Asia Pacific
- China
- India
- Japan
- South Korea
- Oceania
- ASEAN Countries
- Rest of APAC
- Latin America
- Brazil
- Rest of LATAM
- Middle East & Africa
- GCC Countries
- South Africa
- Israel
- Turkey
- Rest of MEA
- North America
Frequently Asked Questions (FAQ) about Preclinical Contract Research Organization (CRO) market report
The market size of preclinical contract research organization (CRO) market was 8.79 billion in 2024.
The market size of preclinical contract research organization (CRO) market is expected to register compound annual growth rate (CAGR) of 12.7% over the forecast period.
Growing reliance on outsourcing by small and virtual biotech, rising demand for specialized services in novel modalities such as gene and cell therapies, oligonucleotides, ADCs, and biologics and advancements in bioanalytical technologies are major key factors driving the market revenue growth of Preclinical Contract Research Organization (CRO) market.
High cost of GLP toxicology and non-human primate studies and shortage of skilled toxicologists and pathologists are key limiting factors driving the market.
Asia Pacific account for fastest revenue growth of 15.9%.
Instruments is the major leading segment of preclinical contract research organization (CRO) market in terms of product.
- Market Definition
- Research Objective
- Research Methodology
- Research Design
- Data Collection Methods
- Primary
- Secondary
- Market Size Estimation
- Top-down Model Type
- Bottom-up Model Type
- Forecasting Methodology
- Tools and Models Used
- Market Overview and Trends
- Market Size and Forecast
- Industry Analysis
- Market Driver, Restraints, Opportunity, and Challenges (DROC) Analysis
- Market Drivers
- Growing reliance on outsourcing by small and virtual biotech
- Rising demand for specialized services in novel modalities such as gene and cell therapies, oligonucleotides, ADCs, and biologics
- Advancements in bioanalytical technologies
- Market Restraints
- High cost of GLP toxicology and non-human primate studies
- Shortage of skilled toxicologists and pathologists
- Market Opportunities
- Implementing AI-driven predictive toxicology and digital platforms
- Expanding services in niche therapy areas such as ophthalmology and rare diseases
- Developing specialized services for viral vector biodistribution, CRISPR off-target analysis, and immunogenicity
- Market Challenges
- Cybersecurity and intellectual property protection concerns
- Regulatory frameworks related to animal welfare, gene editing oversight, and ESG compliance
- Regulatory Landscape
- North America
- Europe
- Asia Pacific
- Latin America
- Middle East and Africa
- Strategic Insights
- Porter’s Five Forces Analysis
- PESTLE Analysis
- Price Trend Analysis
- Value Chain Analysis
- Technological Trends
- Recent Developments
- Funding
- Merger and Acquisition
- Expansion
- Partnership and Collaboration
- Product/ Service Launch
- Product Market Revenue Estimates and Forecasts, 2022-2032
- Instruments
- Microplate readers
- Flow cytometers
- Micro-PET/CT
- Immunohistochemistry (IHC) staining systems
- High-performance liquid chromatography (HPLC) systems
- Mass spectrometers
- Others
- Accessories and Consumables
- Reagents and Chemicals
- Cell Lines
- Antibodies
- Others
- Software
- Instruments
- Service Type Market Revenue Estimates and Forecasts, 2022-2032
- Bioanalysis and DMPK studies
- In vitro ADME
- In-vivo PK
- Toxicology Testing
- GLP
- Non-GLP
- Compound Management
- Process R&D
- Custom Synthesis
- Others
- Chemistry
- Medicinal Chemistry
- Computation Chemistry
- Safety Pharmacology
- Others
- Bioanalysis and DMPK studies
- Model Type Market Revenue Estimates and Forecasts, 2022-2032
- Patient Derived Organoid (PDO) Model
- Patient derived xenograft model
- Application Market Revenue Estimates and Forecasts, 2022-2032
- Oncology
- Neurology
- Cardiology
- Infectious Disease
- Metabolic Disease
- Others
- End-Use Market Revenue Estimates and Forecasts, 2022-2032
- Biopharmaceutical Companies
- Government and Academic Institutes
- Medical Device Companies
- Preclinical Contract Research Organization (CRO) Market Revenue Estimates and Forecasts by Region, 2022-2032, USD Billion
- North America
- North America Preclinical Contract Research Organization (CRO) Market By Product, Market Revenue Estimates and Forecasts, 2022-2032, USD Billion
- Instruments
- Microplate readers
- Flow cytometers
- Micro-PET/CT
- Immunohistochemistry (IHC) staining systems
- High-performance liquid chromatography (HPLC) systems
- Mass spectrometers
- Others
- Accessories and Consumables
- Reagents and Chemicals
- Cell Lines
- Antibodies
- Others
- Software
- Instruments
- North America Preclinical Contract Research Organization (CRO) Market By Service Type, Market Revenue Estimates and Forecasts, 2022-2032, USD Billion
- Bioanalysis and DMPK studies
- In vitro ADME
- In-vivo PK
- Toxicology Testing
- GLP
- Non-GLP
- Compound Management
- Process R&D
- Custom Synthesis
- Others
- Chemistry
- Medicinal Chemistry
- Computation Chemistry
- Safety Pharmacology
- Others
- Bioanalysis and DMPK studies
- North America Preclinical Contract Research Organization (CRO) Market By Model Type, Market Revenue Estimates and Forecasts, 2022-2032, USD Billion
- Patient Derived Organoid (PDO) Model
- Patient derived xenograft model
- North America Preclinical Contract Research Organization (CRO) Market By Application, Market Revenue Estimates and Forecasts, 2022-2032, USD Billion
- Oncology
- Neurology
- Cardiology
- Infectious Disease
- Metabolic Disease
- Others
- North America Preclinical Contract Research Organization (CRO) Market By End-Use, Market Revenue Estimates and Forecasts, 2022-2032, USD Billion
- Biopharmaceutical Companies
- Government and Academic Institutes
- Medical Device Companies
- North America Preclinical Contract Research Organization (CRO) Market Revenue Estimates and Forecasts by Country, 2022-2032, USD Billion
- United States
- Canada
- Mexico
- North America Preclinical Contract Research Organization (CRO) Market By Product, Market Revenue Estimates and Forecasts, 2022-2032, USD Billion
- Europe
- Europe Preclinical Contract Research Organization (CRO) Market By Product, Market Revenue Estimates and Forecasts, 2022-2032, USD Billion
- Instruments
- Microplate readers
- Flow cytometers
- Micro-PET/CT
- Immunohistochemistry (IHC) staining systems
- High-performance liquid chromatography (HPLC) systems
- Mass spectrometers
- Others
- Accessories and Consumables
- Reagents and Chemicals
- Cell Lines
- Antibodies
- Others
- Software
- Instruments
- Europe Preclinical Contract Research Organization (CRO) Market By Service Type, Market Revenue Estimates and Forecasts, 2022-2032, USD Billion
- Bioanalysis and DMPK studies
- In vitro ADME
- In-vivo PK
- Toxicology Testing
- GLP
- Non-GLP
- Compound Management
- Process R&D
- Custom Synthesis
- Others
- Chemistry
- Medicinal Chemistry
- Computation Chemistry
- Safety Pharmacology
- Others
- Bioanalysis and DMPK studies
- Europe Preclinical Contract Research Organization (CRO) Market By Model Type, Market Revenue Estimates and Forecasts, 2022-2032, USD Billion
- Patient Derived Organoid (PDO) Model
- Patient derived xenograft model
- Europe Preclinical Contract Research Organization (CRO) Market By Application, Market Revenue Estimates and Forecasts, 2022-2032, USD Billion
- Oncology
- Neurology
- Cardiology
- Infectious Disease
- Metabolic Disease
- Others
- Europe Preclinical Contract Research Organization (CRO) Market By End-Use, Market Revenue Estimates and Forecasts, 2022-2032, USD Billion
- Biopharmaceutical Companies
- Government and Academic Institutes
- Medical Device Companies
- Europe Preclinical Contract Research Organization (CRO) Market Revenue Estimates and Forecasts by Country, 2022-2032, USD Billion
- Germany
- United Kingdom
- France
- Italy
- Spain
- Benelux
- Nordic Countries
- Rest of Europe
- Europe Preclinical Contract Research Organization (CRO) Market By Product, Market Revenue Estimates and Forecasts, 2022-2032, USD Billion
- Asia Pacific
- Asia Pacific Preclinical Contract Research Organization (CRO) Market By Product, Market Revenue Estimates and Forecasts, 2022-2032, USD Billion
- Instruments
- Microplate readers
- Flow cytometers
- Micro-PET/CT
- Immunohistochemistry (IHC) staining systems
- High-performance liquid chromatography (HPLC) systems
- Mass spectrometers
- Others
- Accessories and Consumables
- Reagents and Chemicals
- Cell Lines
- Antibodies
- Others
- Software
- Instruments
- Asia Pacific Preclinical Contract Research Organization (CRO) Market By Service Type, Market Revenue Estimates and Forecasts, 2022-2032, USD Billion
- Bioanalysis and DMPK studies
- In vitro ADME
- In-vivo PK
- Toxicology Testing
- GLP
- Non-GLP
- Compound Management
- Process R&D
- Custom Synthesis
- Others
- Chemistry
- Medicinal Chemistry
- Computation Chemistry
- Safety Pharmacology
- Others
- Bioanalysis and DMPK studies
- Asia Pacific Preclinical Contract Research Organization (CRO) Market By Model Type, Market Revenue Estimates and Forecasts, 2022-2032, USD Billion
- Patient Derived Organoid (PDO) Model
- Patient derived xenograft model
- Asia Pacific Preclinical Contract Research Organization (CRO) Market By Application, Market Revenue Estimates and Forecasts, 2022-2032, USD Billion
- Oncology
- Neurology
- Cardiology
- Infectious Disease
- Metabolic Disease
- Others
- Asia Pacific Preclinical Contract Research Organization (CRO) Market By End-Use, Market Revenue Estimates and Forecasts, 2022-2032, USD Billion
- Biopharmaceutical Companies
- Government and Academic Institutes
- Medical Device Companies
- Asia Pacific Preclinical Contract Research Organization (CRO) Market Revenue Estimates and Forecasts by Country, 2022-2032, USD Billion
- China
- India
- Japan
- South Korea
- Oceania
- ASEAN Countries
- Rest of Asia Pacific
- Asia Pacific Preclinical Contract Research Organization (CRO) Market By Product, Market Revenue Estimates and Forecasts, 2022-2032, USD Billion
- Latin America
- Latin America Preclinical Contract Research Organization (CRO) Market By Product, Market Revenue Estimates and Forecasts, 2022-2032, USD Billion
- Instruments
- Microplate readers
- Flow cytometers
- Micro-PET/CT
- Immunohistochemistry (IHC) staining systems
- High-performance liquid chromatography (HPLC) systems
- Mass spectrometers
- Others
- Accessories and Consumables
- Reagents and Chemicals
- Cell Lines
- Antibodies
- Others
- Software
- Instruments
- Latin America Preclinical Contract Research Organization (CRO) Market By Service Type, Market Revenue Estimates and Forecasts, 2022-2032, USD Billion
- Bioanalysis and DMPK studies
- In vitro ADME
- In-vivo PK
- Toxicology Testing
- GLP
- Non-GLP
- Compound Management
- Process R&D
- Custom Synthesis
- Others
- Chemistry
- Medicinal Chemistry
- Computation Chemistry
- Safety Pharmacology
- Others
- Bioanalysis and DMPK studies
- Latin America Preclinical Contract Research Organization (CRO) Market By Model Type, Market Revenue Estimates and Forecasts, 2022-2032, USD Billion
- Patient Derived Organoid (PDO) Model
- Patient derived xenograft model
- Latin America Preclinical Contract Research Organization (CRO) Market By Application, Market Revenue Estimates and Forecasts, 2022-2032, USD Billion
- Oncology
- Neurology
- Cardiology
- Infectious Disease
- Metabolic Disease
- Others
- Latin America Preclinical Contract Research Organization (CRO) Market By End-Use, Market Revenue Estimates and Forecasts, 2022-2032, USD Billion
- Biopharmaceutical Companies
- Government and Academic Institutes
- Medical Device Companies
- Latin America Preclinical Contract Research Organization (CRO) Market Revenue Estimates and Forecasts by Country, 2022-2032, USD Billion
- Brazil
- Rest of Latin America
- Latin America Preclinical Contract Research Organization (CRO) Market By Product, Market Revenue Estimates and Forecasts, 2022-2032, USD Billion
- Middle East and Africa
- Middle East and Africa Preclinical Contract Research Organization (CRO) Market By Product, Market Revenue Estimates and Forecasts, 2022-2032, USD Billion
- Instruments
- Microplate readers
- Flow cytometers
- Micro-PET/CT
- Immunohistochemistry (IHC) staining systems
- High-performance liquid chromatography (HPLC) systems
- Mass spectrometers
- Others
- Accessories and Consumables
- Reagents and Chemicals
- Cell Lines
- Antibodies
- Others
- Software
- Instruments
- Middle East and Africa Preclinical Contract Research Organization (CRO) Market By Service Type, Market Revenue Estimates and Forecasts, 2022-2032, USD Billion
- Bioanalysis and DMPK studies
- In vitro ADME
- In-vivo PK
- Toxicology Testing
- GLP
- Non-GLP
- Compound Management
- Process R&D
- Custom Synthesis
- Others
- Chemistry
- Medicinal Chemistry
- Computation Chemistry
- Safety Pharmacology
- Others
- Bioanalysis and DMPK studies
- Middle East and Africa Preclinical Contract Research Organization (CRO) Market By Model Type, Market Revenue Estimates and Forecasts, 2022-2032, USD Billion
- Patient Derived Organoid (PDO) Model
- Patient derived xenograft model
- Latin America Preclinical Contract Research Organization (CRO) Market By Application, Market Revenue Estimates and Forecasts, 2022-2032, USD Billion
- Oncology
- Neurology
- Cardiology
- Infectious Disease
- Metabolic Disease
- Others
- Middle East and Africa Preclinical Contract Research Organization (CRO) Market By End-Use, Market Revenue Estimates and Forecasts, 2022-2032, USD Billion
- Biopharmaceutical Companies
- Government and Academic Institutes
- Medical Device Companies
- Middle East and Africa Preclinical Contract Research Organization (CRO) Market Revenue Estimates and Forecasts by Country, 2022-2032, USD Billion
- GCC Countries
- South Africa
- Israel
- Turkey
- Rest of Middle East and Africa
- Middle East and Africa Preclinical Contract Research Organization (CRO) Market By Product, Market Revenue Estimates and Forecasts, 2022-2032, USD Billion
- North America
- Market Share Analysis
- Revenue Market Share by Key Players (2023-2024)
- Analysis of Top Players by Market Presence
- Competitive Matrix
- Competitive Strategies
- Mergers and Acquisitions
- Partnerships and Collaboration
- Investment and Fundings
- Agreement
- Expansion
- New Product/ Services Launches
- Technological Innovations
- Charles River Laboratories
- Company Overview
- Financial Insights
- Product/ Services Offerings
- Strategic Developments
- SWOT Analysis
- Jubilant Biosys Limited
- Company Overview
- Financial Insights
- Product/ Services Offerings
- Strategic Developments
- SWOT Analysis
- Thermo Fisher Scientific Inc.
- Company Overview
- Financial Insights
- Product/ Services Offerings
- Strategic Developments
- SWOT Analysis
- Clinfinite Solutions
- Company Overview
- Financial Insights
- Product/ Services Offerings
- Strategic Developments
- SWOT Analysis
- Biospective Inc.
- Company Overview
- Financial Insights
- Product/ Services Offerings
- Strategic Developments
- SWOT Analysis
- Altasciences
- Company Overview
- Financial Insights
- Product/ Services Offerings
- Strategic Developments
- SWOT Analysis
- Eurofins Advinus
- Company Overview
- Financial Insights
- Product/ Services Offerings
- Strategic Developments
- SWOT Analysis
- Revvity, Inc.
- Company Overview
- Financial Insights
- Product/ Services Offerings
- Strategic Developments
- SWOT Analysis
- REPROCELL Inc.
- Company Overview
- Financial Insights
- Product/ Services Offerings
- Strategic Developments
- SWOT Analysis
- QPS Holdings
- Company Overview
- Financial Insights
- Product/ Services Offerings
- Strategic Developments
- SWOT Analysis
- Crown Bioscience
- Company Overview
- Financial Insights
- Product/ Services Offerings
- Strategic Developments
- SWOT Analysis
- WuXi AppTec
- Company Overview
- Financial Insights
- Product/ Services Offerings
- Strategic Developments
- SWOT Analysis
- Laboratory Corporation of America Holdings
- Company Overview
- Financial Insights
- Product/ Services Offerings
- Strategic Developments
- SWOT Analysis
- Intertek Group Plc
- Company Overview
- Financial Insights
- Product/ Services Offerings
- Strategic Developments
- SWOT Analysis
- Medpace, Inc.
- Company Overview
- Financial Insights
- Product/ Services Offerings
- Strategic Developments
- SWOT Analysis

